Eye strokes: CRAO, BRVO and other retinal artery and vein occlusions
Schedule an exam
Find Eye DoctorEye strokes occur when blockages (occlusions) occur in arteries or veins in the retina, causing vision loss. The severity of vision loss depends on the extent and location of the occlusion(s) and loss of blood flow.
Just as strokes occur in other parts of the body because blood flow is blocked, your eye also may suffer damage when vital structures such as the retina and optic nerve are cut off from nutrients and oxygen flowing through your blood.
Besides having an eye exam to detect signs of an eye occlusion, you'll also need your family doctor or internal medicine physician to evaluate you for high blood pressure, artery disease or heart problems that may be responsible for the blockage.
If a blockage is found, the type of retinal artery or vein occlusion you have is categorized by its location.
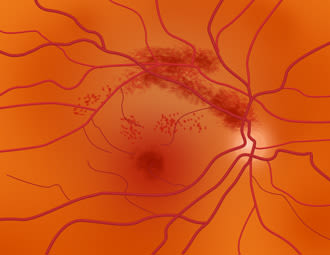
Central Retinal Artery Occlusion (CRAO)
Central retinal artery occlusion usually occurs with sudden, profound, but painless vision loss in one eye. Most people with CRAO can barely count fingers in front of their face or see light from the affected eye.
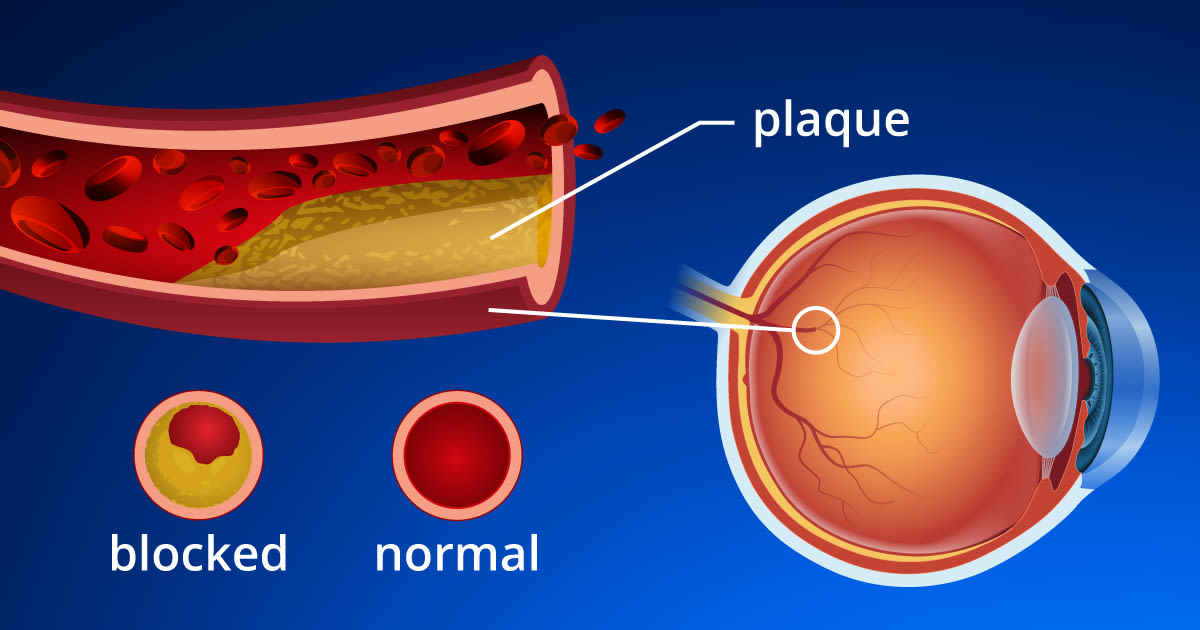
The condition may be preceded by episodes of vision loss known as amaurosis fugax. The cause of CRAO is most commonly a clot or embolus from the neck (carotid) artery or the heart. This clot blocks blood flow to the retina.
CRAO is considered a "stroke" of the eye. Studies show that about two-thirds of patients have underlying high blood pressure and one-fourth of patients will have significant carotid artery disease (plaque with narrowing of the artery lining), cardiac valvular disease or diabetes.
In a recent German study of the underlying risk factors in patients with central retinal artery occlusion, researchers found that previously undiagnosed cardiovascular (CV) risk factors were present in 78 percent of CRAO patients, and 67 percent had CV risk factors in their medical history. The most meaningful unidentified risk factor was narrowing (stenosis) of the carotid artery on the same side of the body as the eye stroke.
Also, 11 of the 84 participants in the study (13 percent) had a stroke either prior to or within one month after diagnosis of CRAO. The study authors concluded a prompt, comprehensive, cardiovascular diagnostic work-up should be considered mandatory for all patients with central retinal artery occlusion.
Your ophthalmologist may diagnose CRAO after an examination of the eye, including a dilated pupil exam. With CRAO the retina will be pale and the vessels narrowed. If you are seen within the first few hours of onset, the retinal signs may not yet be present, and a fluorescein angiogram may be required to confirm the diagnosis. This procedure, which is very safe, entails injection of fluorescein intravenously with retinal photography afterward.
No treatment method has been shown conclusively to benefit CRAO. But if you are seen within 24 hours after acute vision loss begins, many ophthalmologists may attempt to dislodge the embolus through methods such as:
Using glaucoma medications to decrease intraocular pressure.
Having you inhale 5 percent carbon dioxide gas, then using ocular massage.
Performing a minor surgical procedure known as anterior chamber paracentesis, in which numbing drops are used and a small amount of fluid is withdrawn from the front of the eye.
If the embolus can be dislodged, blood flow to the retina may be restored partially. Vision loss is less likely if the occlusion has been present only a very short time. However, studies have shown that the retina suffers irreversible injury after only 90 minutes of blood flow loss (ischemia). Despite all attempts to preserve vision, even when you are seen immediately, most patients suffer severe and permanent visual loss.
Some people with CRAO will have temporal arteritis (giant cell arteritis), an inflammatory condition of the arteries, which calls for treatment with systemic steroids to prevent loss of vision in both eyes.
SEE RELATED: How a stroke can change your vision and Retinal vasculitis
Woodrow Wilson May Have Had An Eye Stroke
Before becoming the 27th U.S. president, Woodrow Wilson woke up one day to almost total blindness in his right eye because of severe bleeding in his retina.

Eye doctors speculate that he had had a central retinal vein occlusion (CRVO), meaning a blockage in the main retinal vein, with resultant bleeding and damage. They base this theory partly on the fact that Wilson had high blood pressure, a risk factor for CRVO.
These days, an eye surgeon would typically treat the area with a laser, to reduce the abnormal blood vessel growth that can occur. But back then, all that Wilson could do was rest his eye for several months. His vision did improve a little over time, though he complained that his golf game was never again as good.
Central Retinal Vein Occlusion (CRVO)
Central retinal vein occlusion (CRVO) causes sudden, painless vision loss that can be mild to severe. Most people will have high blood pressure, chronic open-angle glaucoma and/or significant hardening of the arteries.
For eye occlusion, you may receive ocular massage or glaucoma medications to lower eye pressure.
A study published in February 2013 that was designed to identify risk factors associated with CRVO among patients age 55 and older in the United States found:
African Americans had a 58 percent increased risk of CRVO compared with Caucasians.
Women had a 25 percent decreased risk of CRVO compared with men.
A diagnosis of stroke increased the hazard of CRVO by 44 percent.
A hypercoagulable state (blood clotting disorder) was associated with a 145 percent increased risk of CRVO.
Patients with diabetes or hypertension with end-organ damage had a 92 percent and 53 percent increased risk for CRVO, respectively.
The study authors concluded that high blood pressure and vascular diseases are important risk factors for central retinal vein occlusion, and that blacks have a significantly greater risk for CRVO than other races.
Also, people with diabetes and end-organ damage (diabetic retinopathy, for example) are at increased risk for CRVO, while those with uncomplicated diabetes are not.
When CRVO occurs, the final outcome may involve a thrombus or clot of the central retinal vein just where it enters the eye. Your eye doctor may find mild to severe hemorrhages and cotton-wool spots in the retina (which can indicate poor or absent blood flow).
Initial vision loss when you first are diagnosed with CRVO is a good indicator of the final visual outcome. That is, the worse the vision initially, the worse the final visual acuity. In fact, in half of people with CRVO, final visual acuity remains within three lines on the eye chart of the first visual acuity measurements taken.
Many people with eye blockages have systemic problems such as hardening of the arteries, high cholesterol and high blood pressure.
Two basic classes of CRVO are:
Ischemic: poor blood flow and accompanying poor vision.
Non-ischemic: much better vision when you are first seen and fewer clinical findings.
The prognosis for non-ischemic CRVO is good. But the ischemic type almost always has vision of 20/100 or worse at first, with a much higher risk of developing complications. People with ischemic CRVO must see the eye doctor often, perhaps every few weeks, so they can be evaluated for signs of neovascularization or abnormal vessel growth in the retina and on the iris.
Neovascularization of the retina or optic nerve may cause bleeding (vitreous hemorrhage), and neovascularization of the iris may result in intractable glaucoma, which means high internal eye pressure that fails to respond to conventional therapy.
Both CRVO conditions, if they develop, typically are treated with laser to the retina (pan-retinal photocoagulation) in attempts to cause regression of the neovascularization.
The SCORE study mentioned above found that intraocular corticosteroid injections may help reduce vision loss in people with CRVO. Patients receiving the injections had 5-fold greater odds of significant recovery of their visual acuity, compared with CRVO patients who did not receive the treatment.
In September 2012, Regeneron Pharmaceuticals announced the FDA approved the company's Eylea (aflibercept) monthly eye injections for the treatment of macular edema following central retinal vein occlusion.
Approval of the treatment was based on the results of two studies that showed 56 and 60 percent of patients with macular edema following CRVO who received monthly Eylea injections gained at least 15 letters of best-corrected visual acuity (BCVA) on a standard eye chart after six months of treatment, compared with 12 and 22 percent of patients who received sham injections during the same period.
At the end of the six-month treatment period, patients receiving the Eylea injections gained an average of 17.3 and 18.0 letters of BCVA from baseline visual acuity at the start of the studies, compared with gains of 4.0 and 3.3 letters among patients in the groups that received sham injections.
Eylea previously was granted FDA approval as a treatment for wet macular degeneration in the U.S. in November 2011.
Other treatments for macular edema following central retinal vein occlusion include intraocular injections of Ozurdex (Allergan) or Lucentis (Genentech).
As to the prevalence of retinal vein occlusions (both BRVO and CRVO), a study published in February 2010 that pooled data from population studies from the U.S., Europe, Asia and Australia found:
The prevalence of BRVO was 4.4 per 1,000.
The prevalence of CRVO was 0.8 per 1,000.
Prevalence of all retinal vein occlusions (RVO) varied by race/ethnicity and increased with age, but did not differ by gender.
Hispanics had the highest risk of RVO (6.9 per 1,000), followed by Asians (5.7), African Americans (3.9) and Caucasians (3.7).
Prevalence of CRVO was lower than that of BRVO in all ethnic populations.
Based on the study data, the study authors estimated that 16.4 million adults are affected by retinal vein occlusions, with 2.5 million affected by CRVO and 13.9 million affected by BRVO.
If you have sudden vision loss or any other symptoms of eye stroke, visit your doctor immediately.
SEE RELATED: What is hypertensive retinopathy
Branch Retinal Artery Occlusion (BRAO)
A branch retinal artery occlusion usually occurs suddenly. While typically painless, a BRAO can cause an abrupt loss of peripheral vision. In some cases, you may also lose central vision.
If you have sudden vision loss or other symptoms of an "eye stroke," see your doctor immediately.
Usually the cause is a clot or plaque (embolus) that breaks loose from the main artery in the neck (carotid) or from one of the valves or chambers in the heart.
No ocular therapy has been proven to help. However, some ophthalmologists may try ocular massage or a fluid tap from the eye (anterior chamber paracentesis) in the case of an acute or sudden arterial occlusion. Your ophthalmologist also may prescribe a glaucoma medication to dislodge the embolus, if the condition has been present for fewer than 12 to 24 hours.
Loss of visual acuity with a BRAO will depend mostly on whether arterial blood flow has been disrupted and/or if swelling is present in the macula, where fine focusing occurs.
You also will be evaluated for cardiovascular risk factors and treated accordingly, often in conjunction with your regular doctor.
Most people with BRAO have narrowing of the carotid or neck artery, high blood pressure, cholesterol disorders, cardiac disease or combinations of these disorders.
Your eye doctor will evaluate you every one to two months until your vision is stable. Vision recovery depends on whether the central macula is involved initially.
More than 80 percent of people who have BRAO will recover visual acuity of 20/40 or better*, although most people will have noticeable and permanent vision problems such as blind spots or distortions.
Rarely, you may develop other complications from BRAO, such as neovascularization of the retina or iris. Neovascular glaucoma also is possible.
Branch Retinal Vein Occlusion (BRVO)
People who have a branch retinal vein occlusion near the retina may have decreased vision, peripheral vision loss, distorted vision or blind spots. A BRVO involves only one eye and usually develops in a person with high blood pressure or diabetes.
The cause of BRVO is a localized clot (thrombus) development in a branch retinal vein due to hardening of the arteries (arteriosclerosis) in an adjacent, small branch retinal artery.
Your ophthalmologist will see retinal bleeding along the involved retinal vein in a clear pattern that nearly always leads to the correct diagnosis. Many ophthalmologists will do a fluorescein angiogram during the recovery period if neovascularization is suspected.
A fluorescein angiogram is a safe, in-office diagnostic procedure in which fluorescein dye is administered through the vein (IV) or sometimes orally for retinal photography.
BRVO patients are typically re-evaluated every one to two months to determine if chronic macular swelling (edema) and/or neovascularization are present. If macular edema persists beyond three to six months and visual acuity is reduced below 20/40, you may receive laser treatment.
If you meet guidelines for treatment, laser photocoagulation has been shown to improve vision and increase your chances that final visual acuity will be 20/40 or better. If neovascularization develops or if the BRVO involves a significantly large area of retina leading to neovascularization, you may undergo pan-retinal laser photocoagulation to repair damaged areas.
For many people, retinal hemorrhage and macular swelling will end in a few months, with retention of good vision. If you need laser treatment, your ophthalmologist will use strict criteria to determine whether you will benefit.
These criteria result largely from the Branch Retinal Vein Occlusion Study, in which patients with BRVO who had laser treatment were compared with those who didn't have it.
For macular edema caused by BRVO, your eye doctor may recommend treatment with injections of medication into the eye.
In June 2009, Ozurdex (Allergan) became the first injectable drug therapy to gain FDA approval for the treatment of macular edema following branch retinal vein occlusion or central retinal vein occlusion (CRVO).
Ozurdex treatment consists of injection of a biodegradable implant into the vitreous of the eye that delivers dexamethasone (a potent corticosteroid) to the retina. The implant enables an extended release and effect of dexamethasone to reduce macular swelling and improve visual acuity.
In clinical studies, 20 to 30 percent of patients suffering from retinal vein occlusion experienced a three-line improvement in best-corrected visual acuity with an onset of effect within the first two months following therapy, according to Allergan.
In June 2010, the FDA approved Lucentis (Genentech), another medical treatment for macular edema caused by retinal vein blockage.
Lucentis treatment consists of monthly injections of a medicine called ranibizumab into the vitreous to reduce macular swelling and restore vision. Ranibizumab binds to and inhibits something called vascular endothelial growth factor A (VEGF-A) in the eye, which can trigger the growth of fragile new blood vessels in the retina. These abnormal blood vessels can leak blood and fluid into the eye, contributing to macular edema.
One study leading to FDA approval of Lucentis showed that 61 percent of people treated with monthly ranibizumab injections had significant vision improvement, compared with 29 percent who received sham injections. In another study, 48 percent had significant vision improvement compared with 17 percent who received a placebo.
Lucentis also is approved for the treatment of the wet form of macular degeneration.
READ NEXT: Blood type may increase your risk of some eye disease
Gary Heiting, OD, also contributed to this article.
Cardiovascular risk factors in central retinal artery occlusion: results of a prospective and standardized medical examination. Ophthalmology. September 2015.
A longitudinal analysis of risk factors associated with central retinal vein occlusion. Ophthalmology. February 2013.
Regeneron announces FDA approval of EYLEA (aflibercept) injection for macular edema following central retinal vein occlusion. (Press release issued by the company in September 2012.)
The prevalence of retinal vein occlusion: pooled data from population studies from the United States, Europe, Asia, and Australia. Ophthalmology. February 2010.
A randomized trial comparing the efficiency and safety of intravitreal triamcinolone with observation to treat vision loss associated with macular edema secondary to central retinal vein occlusion: the Standard Care vs Corticosteroid for Retinal Vein Occlusion (SCORE) study report 5. Archives of Ophthalmology. September 2009.
Allergan receives FDA approval for Ozurdex biodegradable, injectable steroid implant with extended drug release for retinal disease. (Press release issued by Allergan in June 2009.)
FDA approves Lucentis (ranibizumab injection) for the treatment of macular edema following retinal vein occlusion. (Press release issued by Genentech in June 2009.)
*Yanoff, Myron and Duker, Jay S. Ophthalmology, Second Edition; Mosby, 2004.
Page published on Thursday, March 14, 2019