What is oxymetazoline hydrochloride?
Oxymetazoline hydrochloride 0.1% is a prescription medication used to non-surgically treat acquired blepharoptosis. A topical ophthalmic solution, oxymetazoline hydrochloride is the first medication approved by the United States Food and Drug Administration (FDA) to temporarily improve acquired ptosis.
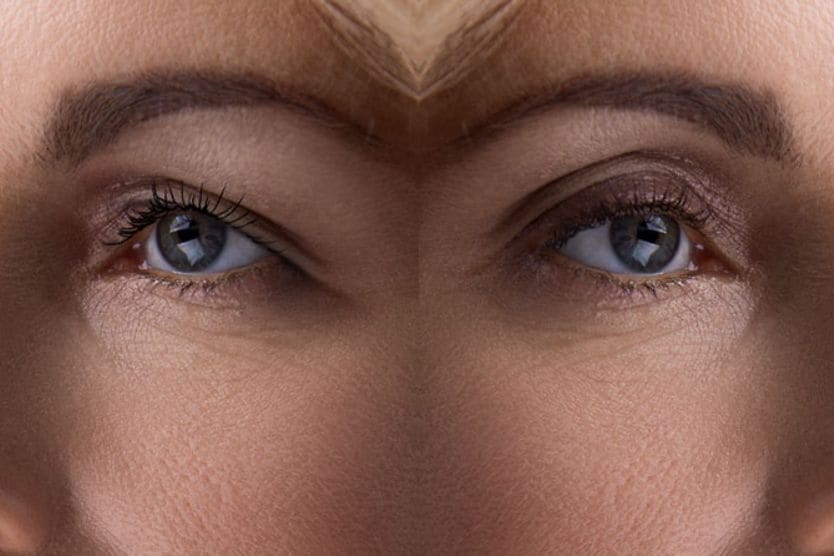
This eyelid condition can affect one or both of the eyes and is characterized by low-lying or droopy upper eyelids. Blepharoptosis is also referred to simply as ptosis (“TOE-sis”).

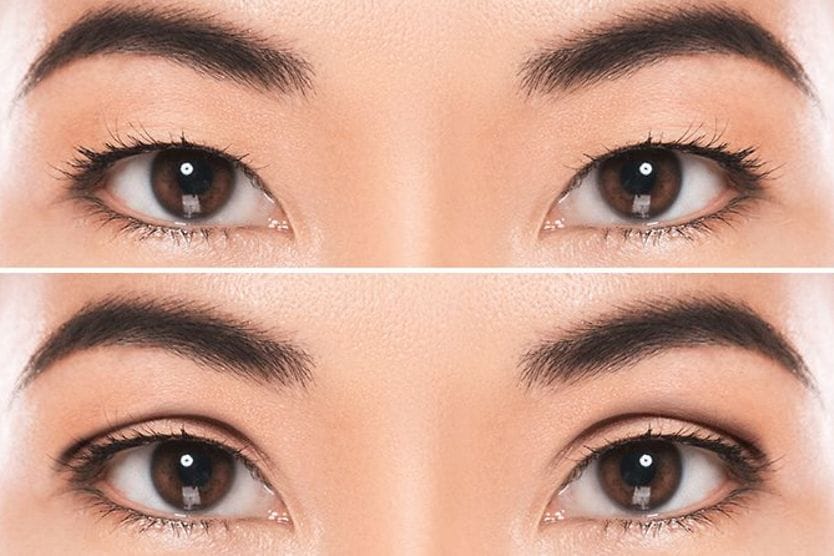
Oxymetazoline is used to treat the condition of droopy eyelids in adults. When administered properly, it can help maintain the raised position of the upper eyelid. It can also contribute to visual field improvement. The results can cause the eye to be more open and look more alert.
It is estimated that approximately 11.5% of adults have ptosis. Low-lying eyelids can have an aesthetic effect on a person’s appearance, making the eyes look hooded and appear tired.
Drooping eyelids can also impair a person’s visual field if the eyelid falls over the pupil of the eye. This may impact their ability to drive, read, use a computer or look upward with an unobstructed view.
Eye conditions treated by oxymetazoline
Ptosis is a common upper eyelid condition. It can be acquired or congenital (present at birth).
Eyelid elevation is controlled by the levator palpebrae superioris (LPS) muscle and the superior tarsal muscle (or Muller's muscle). The levator palpebrae superioris lifts the upper eyelid and keeps it raised.
Muller’s muscle is an involuntary muscle that assists the LPS muscle in maintaining the eyelid in a raised position. When the muscles or the nerves supplying them are damaged, they can fail to function properly. The eyelid can begin to droop, resulting in ptosis.
Oxymetazoline acts on Muller’s muscle by stimulating it to contract, helping to maintain the elevated eyelid. It may be used to treat the following forms of ptosis:
Acquired blepharoptosis
Acquired blepharoptosis can occur at any point in life. It is commonly caused by age or injury. In some cases, ptosis may result following eye treatment or a surgical procedure.
Acquired ptosis can also be caused by:
- Contact lens wear and insertion.
- Surgical instruments used to keep the eye open during eye surgery.
- Other health conditions.
Acquired ptosis may also signal the presence of a serious medical concern. When it occurs in only one eye, ptosis can cause the eyes to appear asymmetrical.
Age-related ptosis
Age is often a reason for droopy eyelids in adults. As people get older, the muscles supporting the upper eyelid can become stretched or weakened. The eyelid skin can begin to stretch and sag as well. This may cause the eyelid to sit lower on the eye.
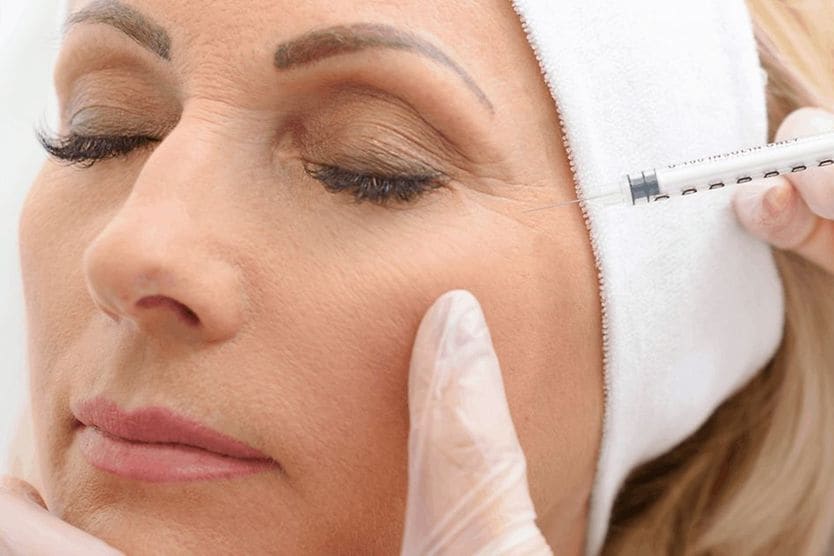
Botulinum toxin-induced ptosis
Ptosis is a common side effect of botulinum toxin injections in the brow area. In these cases, oxymetazoline may be used to improve this effect and treat acquired ptosis due to botulinum toxin injections.
Oxymetazoline hydrochloride before and after
The results of oxymetazoline can vary among individuals. In clinical trials, participants experienced an average of 1mm lift in the upper eyelid with the first use of the medication.
How does it work?
One drop should be placed in one or both eyes. Patients who wear contact lenses should remove the lenses before inserting eye drops and can reinsert them 15 minutes afterwards.
Patients who also use other types of eye drops should wait 15 minutes between applying oxymetazoline and other medications.
How long does it last?
Oxymetazoline must be administered once per day to maintain its effects. In clinical studies, some individuals experienced results within five minutes of receiving the medication. Many experienced results within two hours of administering it. The effects of a single dosage lasted as long as six hours in a number of participants.
Oxymetazoline eye drops can be used long-term.
Who is a candidate?
Ideal candidates for oxymetazoline drops may include adults who have mild to moderate acquired ptosis. Those with age-related droopy eyelids and botulinum toxin- or speculum-induced ptosis may also be candidates for this treatment option.
Oxymetazoline is not designed for patients who have congenital ptosis and has not been tested in children. It is also not intended to treat ptosis due to trauma, nerve issues, eyebrow sagging, or eyelids that droop due to excess skin.
Risks and side effects of oxymetazoline
Oxymetazoline was determined to be safe and well-tolerated when used once daily during clinical trials. These studies took place over the course of 14 to 84 days.
Common side effects were observed in 1%-5% of participants. These included:
- Eye irritation
- Eye redness
- Blurred vision
- Dry eye
- Punctate keratitis
- Headache
- Eye pain (upon instillation)
Women who are pregnant or breastfeeding should consult their physician before using oxymetazoline drops. Individuals with the following conditions should also check with their doctor before using this ophthalmic solution:
- High blood pressure
- Low blood pressure
- Heart conditions
- Cerebral insufficiency
- Coronary artery disease
- Sjögren’s syndrome
- Narrow-angle glaucoma
Patients should also seek medical care if any of their conditions worsen or they develop any of the symptoms outlined above.
Drug interactions may occur with certain medications. These include blood pressure medications, heart medications, monoamine oxidase inhibitors and alpha-adrenergic receptor antagonists. Patients should consult their physician for more information on potential risks, side effects, drug interactions and adverse reactions.
Oxymetazoline vs. surgery
Until recently, the main options for treating droopy eyelids were ptosis surgery and the use of off-label drugs, which could not be used long-term.
The surgical procedure may involve a minor adjustment to the eyelid muscles in some cases. In others, the muscle may need to be strengthened or reattached to allow for eyelid lifting. Surgery carries additional risks and potential side effects, in addition to a recovery period. Surgery is rarely recommended for cases of mild ptosis.
Oxymetazoline offers a potentially long-term nonsurgical alternative to treating acquired ptosis. Individuals may opt for oxymetazoline over blepharoplasty (eyelid surgery) if they are not candidates or are not yet ready for a surgical procedure.
Oxymetazoline hydrochloride vs. botulinum toxin
Botulinum toxin has also served as a nonsurgical option for treating mild cases of droopy eyelids by lifting the eyebrows slightly. It is commonly used to lift the eyebrows and treat fine lines and wrinkles on the upper face.
Sometimes, the use of botulinum toxin for lifting the eyebrow causes drooping of the eyelids because the drug has affected the nerve that controls eyelid muscles. In these cases, oxymetazoline may be recommended to provide a nonsurgical eyelid lift.
READ NEXT: Types of ptosis