Avastin vs. Lucentis: Understanding the differences in anti-VEGF treatments
When Lucentis (ranibizumab) received FDA approval in late June 2006, the new macular degeneration drug was celebrated as a major medical breakthrough.
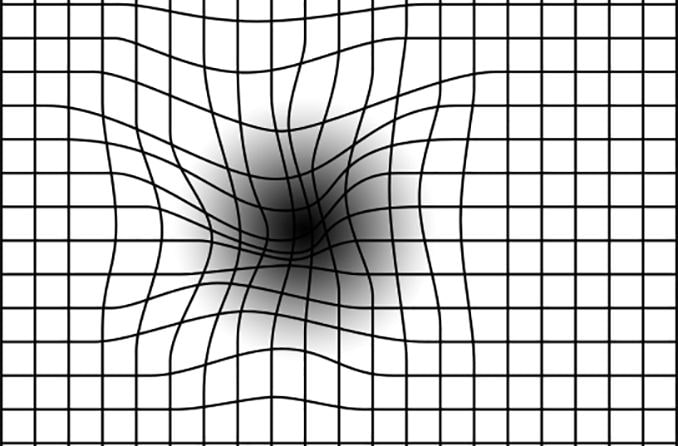
With about 200,000 new cases of advanced, age-related macular degeneration (AMD) identified each year in the United States, many older Americans with more severe or “wet” forms of AMD endured inevitable, gradual loss of central vision.
Now, there is hope for many who once faced certain blindness. Several anti-VEGF drugs have become available for AMD, including four specifically developed for eye conditions:
- Ranibizumab (Lucentis)
- Aflibercept (Eylea and Eylea HD)
- Brolucizumab (Beovu)
- Faricimab (Vabysmo)
Lucentis was the first anti-VEGF drug approved for ophthalmologic use. Clinical trials have shown it can stop and, in many cases, reverse at least some vision loss in most people with advanced AMD. These positive findings clearly make Lucentis one of the most effective, FDA-approved treatments currently available for more damaging forms of AMD.
But some eye doctors argue that a drug closely related to Lucentis, known as Avastin (bevacizumab), has also been shown to be a highly effective and far cheaper alternative for lower-income individuals with advanced AMD. The problem is that Avastin is FDA-approved only for the treatment of colon and other cancers, but not for macular degeneration. As an alternative, many eye doctors have been using Avastin as an off-label treatment.
READ MORE: Anti-VEGF injections: Benefits, risks and what to expect
Genentech initially restricted sales of Avastin for ophthalmic uses
In October 2007, the company that markets both Lucentis and Avastin announced a strategy that was supposed to limit the availability of Avastin for ocular uses.
The company, Genentech, cited safety issues as the reason for halting sales of Avastin to compounding pharmacies that had been dividing Avastin into the smaller quantities needed for treating the eye.
Genentech later responded to widespread protests from eye doctors and organizations, including the American Academy of Ophthalmology (AAO), by announcing that Avastin could still be sold directly to physicians and delivered to destinations of their choice, including compounding pharmacies.
At an emotionally charged AAO conference session in November 2007, eye doctors protested the original decision that they said could have severely reduced supplies of Avastin and deprived lower-income individuals of a sight-saving drug.
Genentech officials stated they would not interfere with a physician’s choice to prescribe Avastin for ophthalmic uses. But while the drug still could be sold to physicians, eye doctors said only compounding pharmacies could deal with sterility issues involved with repackaging Avastin for injection into the eye.
Eye doctors at the AAO conference said they have seen no evidence that the FDA has expressed specific concern about off-label use of Avastin.
Joshua Wenderoff, spokesman for the International Academy of Compounding Pharmacists (IACP), told reporters at the AAO meeting he disputed Genentech’s claim that the decision to stop sales of Avastin to compounding pharmacies was based on safety concerns.
“We believe Genentech is putting profit ahead of patients,” Wenderoff said at the time.
Genentech’s president of product development, Susan Desmond-Hellmann, MD, defended her company’s position, saying that an FDA inspector had asked numerous questions about the propriety of Genentech’s direct sales of Avastin to compounding pharmacies and its off-label use as an ophthalmic drug.
“We stand behind the decision we made,” Desmond-Hellmann said.
Genentech officials stated they worked very closely with any individuals who faced economic hardship from the use of Lucentis, including providing referrals to charitable organizations or other agencies offering assistance.
“Our motive for this is not financially driven,” said Genentech product communications manager Krysta D. Pellegrino. “We do not believe this decision will increase sales of Lucentis. We expect doctors will still have access to Avastin.”
Following the 2007 AAO meeting, Genentech cooperated in the compromise that allowed sales of Avastin directly to eye care physicians who could specify delivery to compounding pharmacies for appropriate formulation needed for treating age-related macular degeneration.
Does Avastin work as well as Lucentis in treating macular degeneration?
Besides cost issues, another area of concern involves which drug works best for treating macular degeneration.
“Tens of thousands of doses of Avastin were given nationwide, while doctors were waiting for ranibizumab [Lucentis] to get approved,” University of Iowa Howard Hughes Medical Institute investigator Edwin M. Stone, MD, PhD, wrote at the time in an editorial published in the October 2006 issue of The New England Journal of Medicine. “And it often worked very well. But what no one knows at the moment is whether one drug is really significantly better than the other.” Since that editorial, multiple studies have demonstrated that Lucentis and Avastin have comparable benefits for vision.
Currently, Lucentis costs about $1,575 per injection, while Avastin costs about $20 to $50. Although the price of Avastin increases after repackaging for ocular use, it still remains lower than that of Lucentis. This price discrepancy could be highly significant for people who have limited or no health insurance coverage.
Medicare covers injectable treatments for wet age-related macular degeneration (AMD), including both Avastin (bevacizumab) and Lucentis (ranibizumab), under Medicare Part B. After you meet the Part B deductible, you pay 20% of the Medicare-approved amount for the drug and your doctor’s visit.
An investigation published in 2023 in JAMA points out that the 20% co-payment required for each monthly injection still represents a significant expense. Supplemental insurance might be available to defray at least some costs involved with co-payments.
However, Avastin may still be the cheaper alternative, even for people covered by Medicare or health insurance.
Additionally, more affordable versions of Lucentis, known as biosimilars, have become accessible in numerous countries. These medications are equally effective as the original Lucentis but can come at a lower cost, typically ranging from 30% to 80% of the original price. Avastin is often still more cost-effective than biosimilars. However, as the availability of biosimilars increases, they have the potential to expand access to treatment for eye conditions.
More about Lucentis and Avastin
Both Lucentis and Avastin are produced by the same company, Genentech, based in San Francisco, CA. But there are differences between the two drugs.
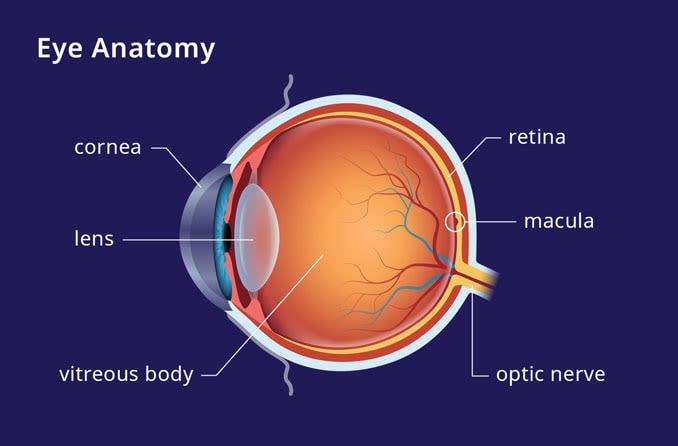
Lucentis is administered in the form of smaller molecules, which is thought to give Lucentis an advantage over Avastin in its ability to penetrate the eye’s retina and halt abnormal blood vessel growth contributing to advanced macular degeneration and scarring that causes blindness.
Genentech company officials have repeatedly told news reporters that considerable expense was involved in developing Lucentis as a macular degeneration treatment and in funding clinical trials proving the drug’s safety and effectiveness. They indicated that the company had no intention of also funding clinical trials for Avastin as a treatment for macular degeneration, now that Lucentis has FDA approval, and the need for an effective macular degeneration treatment has been met. Instead, U.S. government funds were used to compare the effectiveness and safety of the two different treatments.
In early 2008, plans were announced for enrollment of participants in the two-year Comparison of Age-Related Macular Degeneration Treatments Trials (CATT), sponsored by the National Eye Institute at 44 study sites. The study found that both drugs had similar outcomes. Since then, many studies have demonstrated that Avastin is effective and safe for treating eye disease.
Where the Lucentis-Avastin debate now stands
In the past, serious safety concerns were expressed about off-label use of Avastin as a macular degeneration treatment. This is partly because the FDA in January 2005 warned that Avastin, when used to treat colon and other cancers, may significantly increase the risk of stroke, heart attack and other related adverse health events.
However, current evidence shows that when used for eye treatments, bevacizumab and ranibizumab show no significant differences in safety profiles. This is likely because a relatively small amount of the drug is injected into the eye, compared with the higher doses needed for systemic treatment.
The debate was largely settled when the CATT clinical trials comparing the two treatments were completed. The CATT study showed similar outcomes for Avastin and Lucentis after two years of treatment for wet AMD.
Over the years, studies have continued to find that Lucentis and Avastin result in similar improvements in vision. These studies also demonstrate a comparable safety profile. A major advantage of Avastin is its lower cost per injection compared to Lucentis. The final difference in price, however, will be determined by insurance. Ultimately, both drugs have helped millions of people keep their vision and stay independent.