Understanding what keratoconus is and how it’s treated
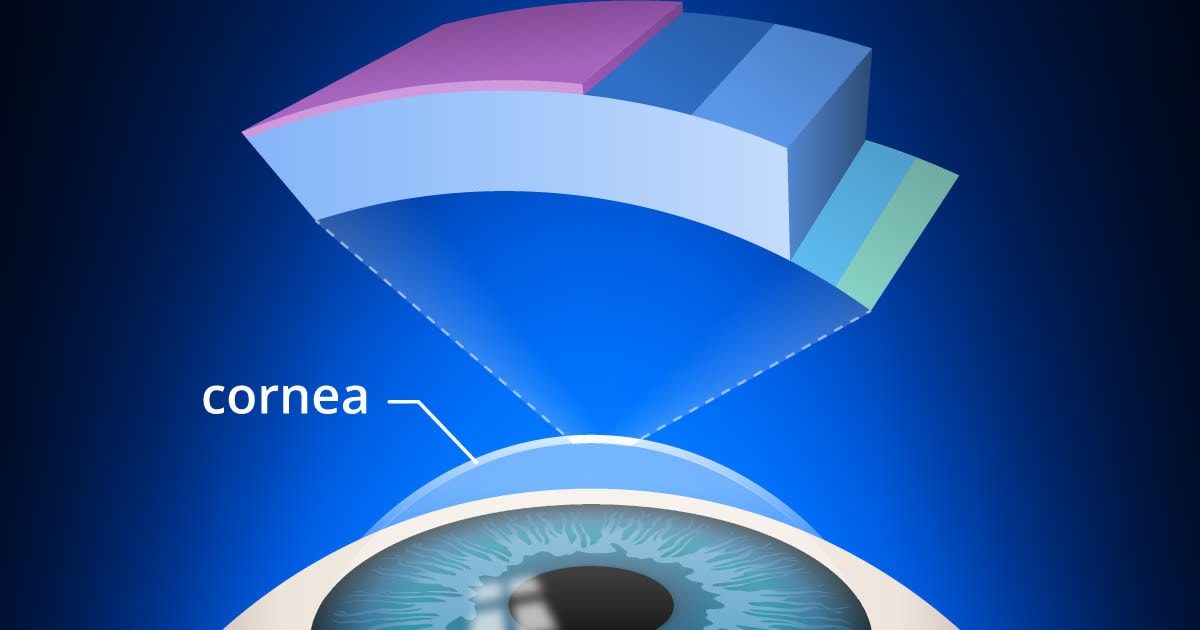
Keratoconus is a progressive eye disease in which the normally round cornea thins and begins to bulge into a cone-like shape. This cone shape deflects light as it enters the eye on its way to the light-sensitive retina, causing distorted vision.
Keratoconus can occur in one or both eyes and often begins during a person’s teens or early 20s. Without treatment, it can lead to irreversible vision loss. Although the condition is generally not preventable, treatment can help preserve vision.
Keratoconus signs and symptoms
As the cornea becomes more irregular in shape, it causes progressive myopia and irregular astigmatism to develop. This creates additional problems with distorted and blurred vision. Symptoms such as glare and light sensitivity may also occur.
Often, keratoconic patients experience changes in their eyeglass prescription from one eye doctor visit to the next.
What causes keratoconus?
Research suggests that the weakening of the corneal tissue leading to keratoconus may be due to an imbalance of enzymes within the cornea. This imbalance makes the cornea more susceptible to oxidative damage from compounds called free radicals, causing it to weaken and bulge forward.
Risk factors for oxidative damage and weakening of the cornea include a genetic predisposition. This helps explain why keratoconus often affects more than one member of the same family.
Keratoconus is also associated with:
- Overexposure to ultraviolet (UV) rays from the sun
- Excessive eye rubbing
- A history of poorly fitted contact lenses
- Eye allergies and chronic eye irritation
Keratoconus treatment
In the mildest form of keratoconus, eyeglasses or soft contact lenses may help. But as the disease progresses and the cornea thins and becomes more irregular in shape, glasses and regular soft contact lens designs no longer provide adequate vision correction.
Treatments for progressive keratoconus include:
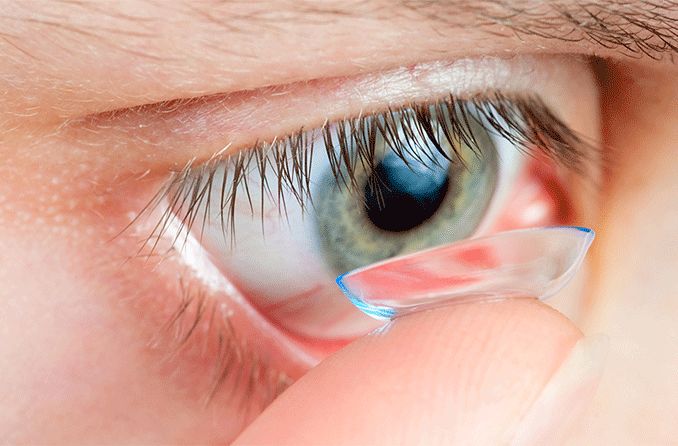
1. Custom soft contact lenses
Contact lens manufacturers have introduced custom soft contact lenses specially designed to correct mild-to-severe keratoconus. These lenses are made-to-order based on detailed measurements of the person’s keratoconic eye(s). They may be more comfortable than other types of contact lenses for some wearers.
Custom soft contacts available for the treatment of keratoconus include options such as:
- Medium-water hydrogel lenses can correct up to 30 D of nearsightedness or farsightedness and up to -10 D of astigmatism.
- High-water silicone hydrogel lenses can correct up to 30 diopters (D) of myopia (nearsightedness) or hyperopia (farsightedness) and up to -15 D of astigmatism.
Both lenses have a wide range of fitting parameters for a customized fit. They’re larger in diameter than regular soft lenses, providing greater stability on a keratoconic eye.
Custom soft contacts are typically more expensive than regular soft contacts. However, vision insurance may cover some of the costs associated with contact lenses for keratoconus.
2. Gas permeable (GP) lenses
Glasses and soft contacts often cannot provide adequate visual acuity in cases of keratoconus. Because of this, gas permeable contacts are usually the preferred treatment. GP lenses vault over the cornea, replacing its irregular shape. This provides a smooth, uniform refracting surface to improve vision.
Fitting specialty contact lenses on an eye with keratoconus often is challenging and time-consuming. Patients likely need frequent visits to their eye doctor to fine-tune the fit and prescription. This is especially so if keratoconus continues to progress.
3. “Piggybacking” lenses
Fitting a GP lens over a cone-shaped cornea can sometimes be uncomfortable for a person with keratoconus. In such cases, some eye doctors advocate a technique called “piggybacking.” This involves wearing two different types of lenses on the same eye.
For keratoconus, a soft contact (such as one made of silicone hydrogel) is placed over the eye. A GP lens is then fitted over the soft lens. This approach increases wearer comfort because the soft lens acts like a cushion under the rigid GP lens.
Eye doctors will closely monitor the fitting of “piggyback” contacts to make sure enough oxygen reaches the surface of the eye. (This can be a problem when two lenses are worn on the same eye.) However, many modern contacts — both GP and soft — typically have adequate oxygen permeability for a safe “piggyback” fit.
4. Hybrid lenses
Hybrid contacts are a blend of a GP and a soft contact lens. Certain hybrid contacts combine a highly oxygen permeable rigid center with a soft peripheral “skirt.” The central GP zone of the lens vaults over the cone-shaped cornea for increased comfort. These lenses were designed specifically for keratoconus.
Hybrid contacts provide the crisp optics of a gas permeable contact lens and wearing comfort that rivals that of soft lenses. Some of these lenses are available in a wide variety of parameters to provide a fit that conforms well to the irregular shape of a keratoconic eye.
5. Scleral and semi-scleral lenses
These are large-diameter GP lenses. They’re large enough that the periphery and edge of the lens rest on the sclera (the white of the eye) and the conjunctiva (the clear tissue lining the inner eyelids and front of the eye).
Scleral lenses cover a larger portion of the sclera, whereas semi-scleral lenses cover a smaller area.
The center of scleral and semi-scleral lenses vaults over the irregularly shaped cornea. As a result, these lenses don’t apply pressure to the eye’s cone-shaped surface. This design provides a more comfortable fit.
These larger lenses are also more stable than conventional GP contacts. Because conventional options cover only part of the cornea, they tend to move with each blink.
6. Prosthetic lenses
Eyes with advanced keratoconus may require an advanced prosthetic scleral lens design.
One example is lenses made with a custom impression-based scleral contact lens system.This system uses proprietary technology to match the unique irregularities of each eye.
The process begins with taking an impression mold. The impression captures the precise curvatures of the entire ocular surface. Doctors then send the impression to a lab for scanning.
The scanned data is fed to a numerically controlled machining system to produce a lens that matches the impression. It provides an exact match, fitting the individual cornea and sclera perfectly. The precise design of the back surface allows high-quality, customized optics to be placed on the front surface of the device.
Only eye doctors who undergo specific training and certification can fit these custom scleral lenses.
7. Corneal cross-linking (CXL)
This procedure, also called corneal collagen cross-linking, strengthens the cornea to prevent further bulging. It does this by creating additional bonds (“anchors”) between collagen fibers.
There are two versions of corneal cross-linking:
- Epithelium-off – The outer layer of the cornea (the epithelium) is removed to allow the entry of riboflavin, a type of B vitamin, into the cornea. The riboflavin is then activated with UV light. Benefits include a more rigid and flatter cornea and a reduced risk of disease progression.
- Epithelium-on – The corneal epithelium is left intact during the treatment. Advantages include a reduced risk of infection, less discomfort and one-day visual recovery.
CXL may significantly reduce the need for corneal transplants among keratoconus patients.
8. Intrastromal corneal ring segments
Intrastromal corneal ring segments are FDA-approved, clear, arc-shaped corneal inserts. They’re surgically placed in the peripheral cornea to reshape the front surface of the eye for clearer vision.
Intrastromal corneal ring segments may be needed when keratoconus patients can no longer obtain functional vision with contact lenses or eyeglasses.
Studies have shown that intrastromal corneal ring segments can improve the best spectacle-corrected visual acuity (BSCVA) of a keratoconic eye by an average of two lines on a standard eye chart. The implants also have the advantage of being removable and exchangeable. The surgical procedure only takes about 10 to 20 minutes to complete.
Intrastromal corneal ring segments might delay the need for a corneal transplant. However, they cannot always prevent one if keratoconus continues to progress.
9. Corneal transplant
Some people with keratoconus can’t tolerate a rigid contact lens. Others may reach a point where contacts or alternative therapies no longer provide acceptable vision. In these cases, a corneal transplant (keratoplasty) may be considered.
Different types of corneal transplants are used to treat keratoconus, including:
- Penetrating keratoplasty (PKP) – This replaces the entire cornea. It’s also called a full-thickness corneal transplant.
- Deep anterior lamellar keratoplasty (DALK) – This replaces the front and middle corneal layers while leaving the back layers intact.
- Anterior limiting lamina transplantation – This replaces the front layer of the cornea.
Even after a transplant, glasses or contacts are often needed for clear vision.
10. Corneal tissue addition for keratoplasty (CTAK)
This is a newer keratoconus treatment that offers an alternative to corneal transplants for some people. It involves adding custom-sized areas of donor tissue to reinforce the cornea’s structure and smooth surface irregularities.
CTAK helps improve visual acuity and enables a more precise, comfortable fit for contact lenses. It was launched in 2024 and is not yet FDA-approved.
SEE RELATED: What is corneal ectasia?
11. Combined treatments
In some cases, keratoconus treatments may be combined to provide enhanced results for corneal health and vision. Combination treatments may include:
Topography-guided photorefractive keratectomy (PRK) with corneal cross-linking (CXL)
Also called the “Athens Protocol,” this method combines photorefractive keratectomy (PRK) and CXL. PRK is a procedure that reshapes the cornea, while CXL helps keep it from bulging.
Intrastromal corneal ring segments (ICRS) with corneal cross-linking (CXL)
Using a combination of CXL and intrastromal corneal ring segments has also shown promising results for treating keratoconus. CXL reduces the bulging of the cornea, while intrastromal corneal ring segments can create a flatter, smoother corneal surface.
Corneal cross-linking (CXL) with toric phakic intraocular lens (pIOL)
Progressive mild-to-moderate cases have been treated with the combination of CXL and implantation of a toric phakic intraocular lens (IOL). CXL is first performed to stabilize the cornea. Once the cornea is stable, a pIOL is then implanted to correct astigmatism or myopia caused by the condition.
Penetrating keratoplasty (PKP) with corneal cross-linking (CXL)
This involves a combination of PKP (replacing the entire cornea) with CXL to strengthen it and prevent the grafted tissue from bulging. (PKP tissue can be treated before or after transplantation.)
Penetrating keratoplasty (PKP) with specialty lenses
Many patients still need vision correction with custom soft contact lenses and specialty lenses following PKP (a corneal transplant). The ideal solution can be a soft contact lens, a rigid gas permeable lens (RGP) or a combination of the two (called a “piggyback” combination, where a soft contact lens is worn underneath an RGP lens).
Corneal transplant with cataract surgery and artificial lens implementation
When both keratoconus and a cataract are present, eye doctors may recommend a triple procedure that includes:
- Corneal transplant with PKP – Replacing the entire cornea
- Cataract removal – Removing the clouded natural lens from the eye
- Posterior chamber intraocular lens (PCIOL) implementation – Inserting a clear artificial lens to replace the clouded lens and improve vision
This combination of procedures addresses multiple concerns within one surgery.
Consult your eye doctor about keratoconus
Keratoconus is a progressive eye condition affecting the shape of the cornea. The changes it causes can impact your vision over time. However, the condition can be effectively managed through a range of treatment options.
If you experience blurry, distorted vision or other keratoconus symptoms, it is recommended to seek professional care right away. Early diagnosis and treatment may help preserve your vision and reduce the risk of disease progression.