Narrow-angle glaucoma (angle-closure glaucoma)
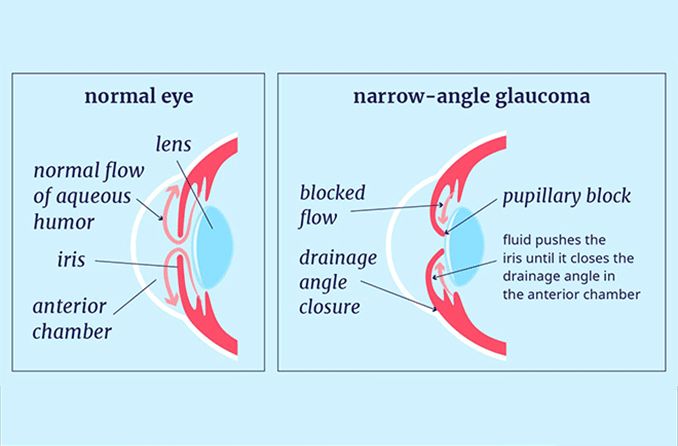
Narrow-angle glaucoma is a type of glaucoma that occurs when the structure inside the eye that allows fluid to drain normally from the eye (called the drainage angle) becomes restricted.
Narrow drainage angles in the eye increases the risk of the drainage angle becoming blocked, resulting in a significant increase in eye pressure and risk of vision loss. If this occurs, narrow-angle glaucoma is usually called angle-closure glaucoma.
Closure of the drainage angle can occur gradually, partially and intermittently with few symptoms (chronic angle-closure glaucoma) or suddenly and severely with painful symptoms (acute angle-closure glaucoma). Acute angle-closure glaucoma is a medical emergency and requires immediate attention.
Causes of narrow-angle glaucoma
Generally, narrow-angle glaucoma is caused by the iris (the colored part of the eye that surrounds the pupil) being bowed forward, restricting the space between the clear cornea and the iris where the drainage angle of the eye is located.
Conditions associated with narrow-angle glaucoma include:
Iris plateau – In this condition, the ciliary body (the structure behind the iris that produces the aqueous fluid in the eye) is positioned too far forward in the eye. This displaces the peripheral iris forward, causing the drainage angle to be narrower than normal and at greater risk for obstruction by the front surface of the peripheral iris.
Hyperopia – People who are highly farsighted are more likely to have eyes with shallow anterior chambers (the space in the front of the eye, between the cornea and iris) and narrow angles, increasing their risk for angle-closure glaucoma.
Tumors and other causes – A tumor behind the iris, swelling associated with inflammation of the ciliary body (intermediate uveitis) and alteration of the shape of the eye after surgery for a detached retina also can cause angle-closure glaucoma.
Narrow-angle glaucoma symptoms
While narrow-angle glaucoma is uncommon, when it occurs, it can cause much more rapid and severe increases in eye pressure and vision loss than the much more common form of glaucoma called primary open-angle glaucoma. This is especially true if the drainage angle closes suddenly, as it does in the acute angle-closure form of narrow angle glaucoma.
Therefore, it’s critical to be aware of the signs and symptoms of this particularly dangerous type of narrow-angle glaucoma.
The Glaucoma Research Foundation lists these signs and symptoms of the acute angle-closure variety of narrow-angle glaucoma:
Sudden severe eye and head pain
The appearance of rainbow-colored circles around bright lights
Hazy or blurred vision
Sudden loss of vision
Other symptoms include abnormally dilated pupils, red eyes, nausea and vomiting.
The duration for which you experience these symptoms varies. However, regardless of how long symptoms persist, each angle-closure episode potentially results in partial loss of your peripheral vision.
For this reason, acute angle-closure glaucoma is considered a medical emergency. If the high eye pressure is not reduced within hours, it can cause permanent vision loss. Anyone who experiences these symptoms should immediately contact an ophthalmologist or go to a hospital emergency room.
SEE RELATED: Glaucoma and headaches
Risk factors for narrow-angle glaucoma
There are certain characteristics that put an individual at higher risk for narrow-angle glaucoma. These risks include:
Hyperopia – People who are farsighted are more likely to have eyes with shallow anterior chambers and narrow angles, increasing their risk for narrow-angle glaucoma.
Age – As you grow older, the anterior chamber tends to become increasingly shallow, and the drainage angle may narrow as you age.
Race – Anatomically, individuals who are Asian, as well as Inuit and other northern indigenous people, have narrower anterior chamber drainage angles. This results in a higher incidence of narrow-angle glaucoma in these populations.
Sex – When studied in Caucasians, narrow-angle glaucoma occurs three times more frequently in women than in men. Among African Americans, men and women appear to be affected equally.
Narrow-angle glaucoma treatments
The best way to treat narrow-angle glaucoma is to keep your eye pressure at a safe, stable level. Some ways to achieve this include:
Eye drops – Topical eye drops for glaucoma may be sufficient to treat some cases of narrow-angle glaucoma.
Surgery – If signs of narrow-angle glaucoma are detected during an eye exam, your eye doctor may suggest glaucoma surgery to reduce your risk of angle-closure attacks.
Procedures called iridotomy and iridectomy are typically performed on narrow-angle glaucoma patients.
During an iridotomy, surgeons use a laser to create a small hole in the peripheral iris to increase fluid drainage and decrease the risk of angle closure. An iridectomy involves removing a small piece of the iris to make better fluid flow in the eyes possible.
Awareness of narrow-angle glaucoma’s potential sudden onset and severe risk can easily cause unrest, but don’t worry. The disease is easily detectable in a comprehensive eye exam and is treatable by a variety of surgical and non surgical options.
Knowledge is the first step in prevention. If you fall under “at-risk” and it’s been a while since your last eye exam, it’s time to see your eye doctor. At the very least, you gain peace of mind. At most, you save your vision.
SEE RELATED: Natural remedies for glaucoma
Page published on Thursday, July 23, 2020






