Corneal cross-linking is a minimally invasive outpatient procedure designed to treat progressive keratoconus (and, sometimes, other conditions that cause a similar weakening of the cornea).
The corneal cross-linking procedure strengthens and stabilizes the cornea by creating new links between collagen fibers within the cornea. The two-step procedure applies specialty formulated riboflavin (vitamin B) eye drops to the surface of the eye immediately followed by a controlled exposure of the eye to ultraviolet light.
The two basic types of corneal cross-linking are:
- Epithelium-off cross-linking. In this procedure, the thin outer layer (epithelium) of the cornea is removed to allow the liquid riboflavin to more easily penetrate the deeper corneal tissue.
- Epithelium-on cross-linking. In this procedure (also called transepithelial cross-linking), the protective corneal epithelium is left intact, making it a less invasive procedure than cross-linking with epithelium removal.
Corneal cross-linking can be combined with other procedures for keratoconus treatment. For example, it can be performed along with implantation of tiny arc-shaped corneal inserts called Intacs to reshape and stabilize the cornea in more advanced cases of keratoconus.
Currently, the only corneal cross-linking platform that is FDA-approved for the treatment of progressive keratoconus in the U.S. is epithelium-off cross-linking performed with a Glaukos KXL System and proprietary Photrexa and Photrexa Viscous solutions.
The use of any drug or drugs other than Photrexa and Photrexa Viscous or any device other than the KXL System for corneal cross-linking is not FDA approved, except as part of an investigational new drug study.
Glaukos has completed patient enrollment in a Phase 3 clinical trial to evaluate the safety and efficacy of an epithelium-on (epi-on) corneal cross-linking procedure for the treatment of progressive keratoconus.

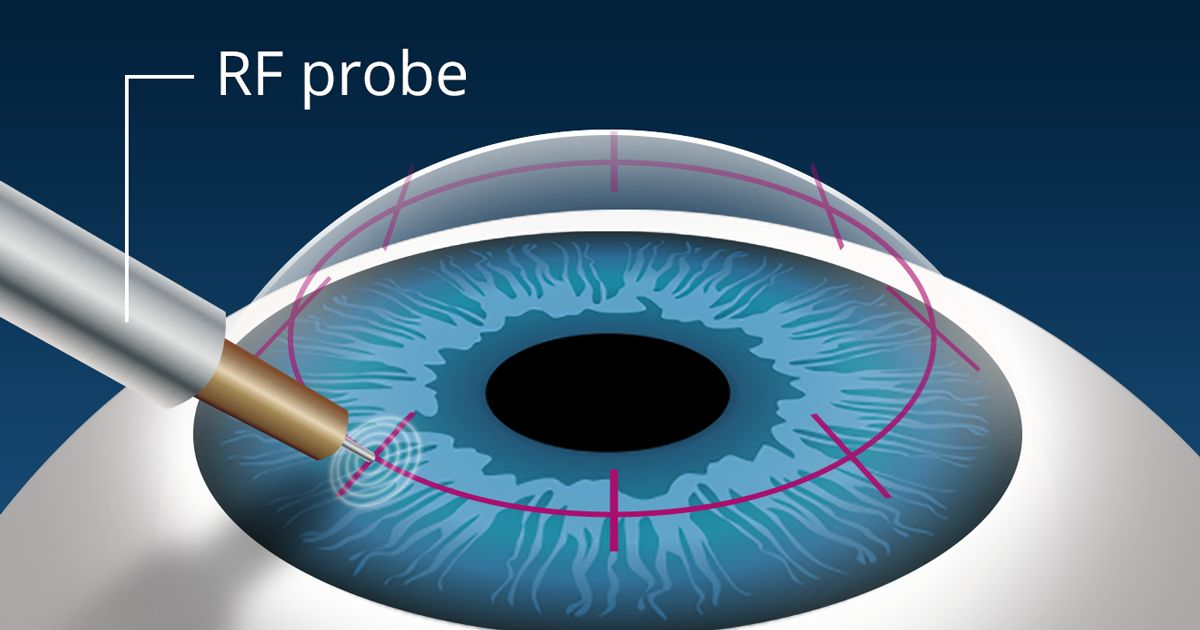
Corneal cross-linking (CXL) strengthens a weak or thin cornea with a combination of riboflavin solution and controlled UV light. (Image: Glaukos, Inc.)
Best candidates for corneal cross-linking
Corneal cross-linking is most effective if it can be performed before the cornea has become too irregular in shape or there is significant vision loss from keratoconus. If applied early, cross-linking typically will stabilize or even improve the shape of the cornea, resulting in better visual acuity and an improved ability to wear contact lenses.
Other potential applications of cross-linking include the treatment of corneal ulcers that are unresponsive to treatment with topical antibiotics. Studies also have found that corneal cross-linking can be effective for eradicating a variety of corneal infections.
Some surgeons have also reported reasonable results in improving the stability of the cornea in people who have undergone radial keratotomy (RK), an incisional refractive procedure from the 1980s and 1990s. Cross-linking appears to work best for those who are experiencing daily fluctuations as a result of their RK procedures.
People who are considering vision correction procedures such as LASIK also might eventually be pre-treated with corneal cross-linking to strengthen the eye's surface before undergoing an excimer laser ablation that reshapes the cornea.
What to expect during and after corneal cross-linking
During a preliminary evaluation, your eye doctor will measure the thickness of your cornea and make sure you are a good candidate for the procedure. You also will need to have a routine eye exam to assess your vision and general eye health.
Detailed mapping of the shape of your cornea (called corneal topography) will also be performed.

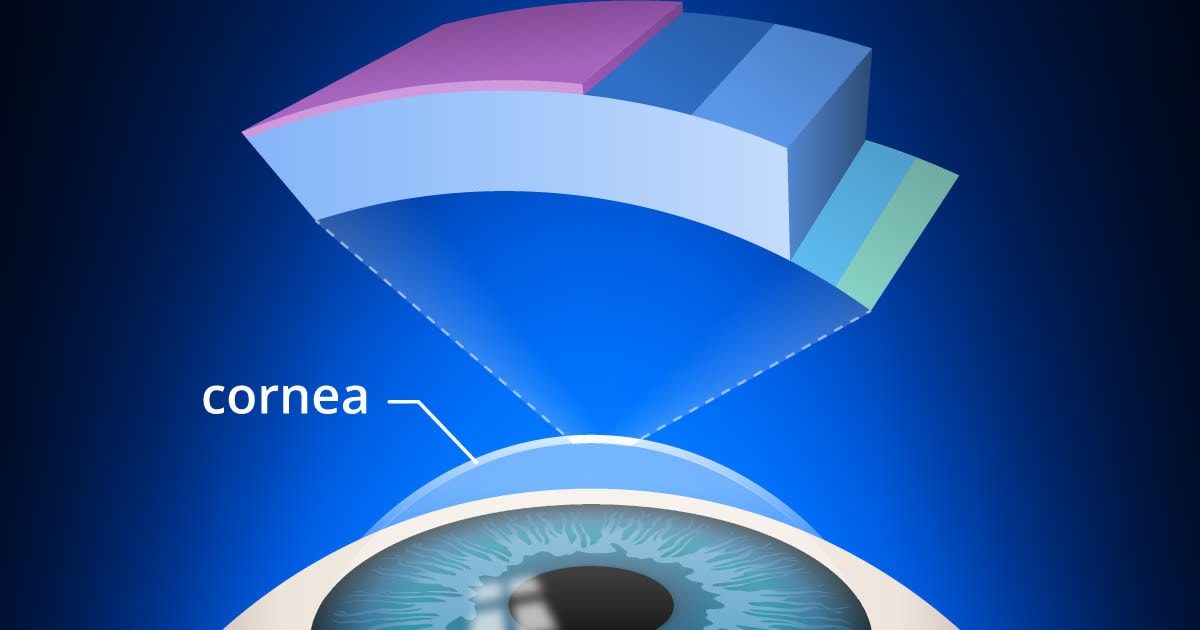
Layers of the cornea. Corneal cross-linking strengthens bonds in the stroma of the cornea, which is the layer from which tissue is removed during LASIK surgery.
The cross-linking procedure takes about an hour in most cases, depending on the technique used.
If you are undergoing an epithelium-off ("epi-off") procedure, your surgeon will place you in a reclining position and remove the epithelial layer from your cornea. Riboflavin eye drops will then be placed on your eye.
If you undergo an epithelium-on ("epi-on") procedure, you are placed in a reclining position, and riboflavin drops are applied. After enough time has passed (depending on the technique), the surgeon may evaluate your eye to ensure that the cornea has absorbed a sufficient amount of riboflavin.
Corneal thickness may be checked, and then the UV light will be applied for up to 30 minutes.
After epi-off and epi-on cross-linking, a bandage contact lens will be placed on your eye to reduce discomfort and facilitate healing. Topical antibiotic and anti-inflammatory drops will be prescribed.
Epi-off cross-linking studies have found that most keratoconus patients will remain stable or achieve improvement in the corneal shape, which is highly significant for people with progressive keratoconus that otherwise might lead to severe vision loss. Epi-on cross-linking is currently under clinical investigation to determine the safety and efficacy of the procedure.
Cost of corneal cross-linking
The cost of corneal cross-linking for the treatment of progressive keratoconus typically ranges from $2,500 to $4,000 per eye.
Insurance coverage for FDA-approved epithelium-off cross-linking with the KXL System and Photrexa drug formulations from Glaukos is widely available. Ask your eye doctor or insurance company for assistance in determining what your total out-of-pocket expense for the procedure will be.