What is aniridia?
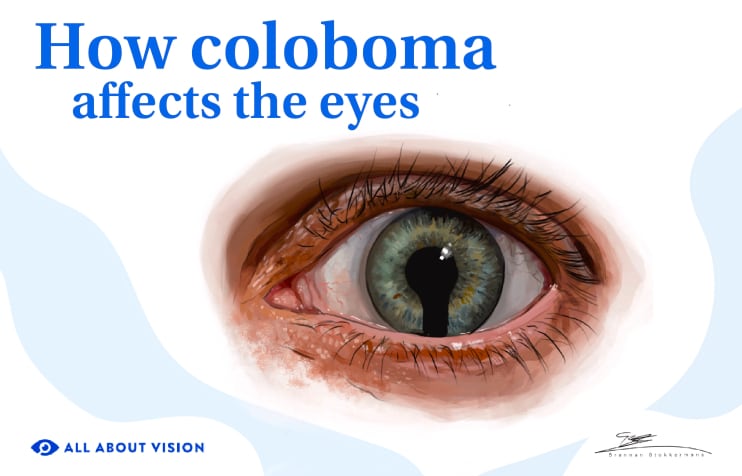
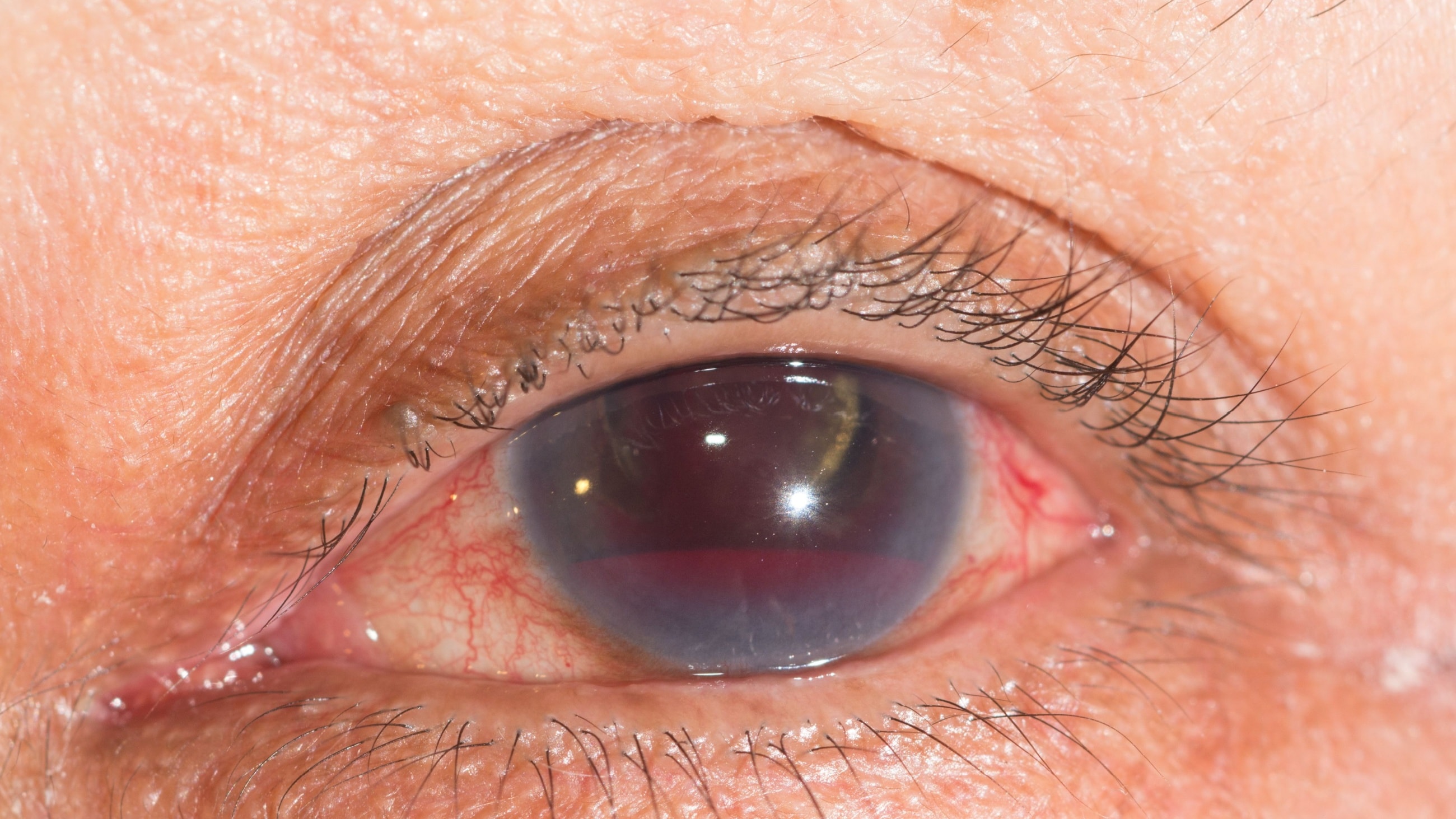
Aniridia is a rare condition that causes babies to be born without an iris (the colored ring around the pupil). The whole iris may be missing or just part of it. Aniridia could also cause other parts of the eye to be underdeveloped. This can lead to additional eye issues and impaired vision.
How common is aniridia?
Aniridia is a rare condition, with estimates ranging from 1 in every 40,000 to 1 in every 100,000 people affected. It does not happen more often in any one gender or ethnicity, but it is more likely to happen if one parent already has aniridia.
What causes aniridia?
Aniridia is congenital, meaning it’s present from birth. While the condition is very uncommon, there is an increased chance of developing if a parent already has aniridia. In these cases, a child has a 50% chance of aniridia, making up two out of three people (one parent and one child) with aniridia. Additionally, sporadic aniridia can occur when the baby has aniridia but the parents do not have the condition.
Aniridia is the result of a gene variant or mutation, which is when there’s an alteration in the DNA sequence that makes up a gene. It’s primarily caused by variants of the PAX6 gene, but there are other genes that might be involved in the disease too. The PAX6 gene is important for the development of several parts of the eye in addition to the iris, including the:
- Retina – The tissue lining in the back of the eye that senses light.
- Cornea – The clear “dome” in the front of the eye.
- Lens – The crystalline structure behind the pupil that helps focus light toward the back of the eye.
The PAX6 gene is also involved in developing other parts of the body (like the central nervous system, spine and pancreas). Changes to the PAX6 gene can affect these other parts, which can cause other conditions to happen alongside aniridia.
What are the signs and symptoms of aniridia?
Without a visible iris, the pupil tends to look bigger. The iris is responsible for adjusting the pupil according to the amount of light, so, without an iris, the pupil might have a harder time adjusting to these changes. This can lead to:
- Blurry vision or low vision
- Partial or complete blindness in one or both eyes
- Light sensitivity
- Strabismus, when one or both eyes do not line up (commonly referred to as a lazy eye)

Possible complications of aniridia
Aniridia can be an isolated condition, or it can lead to other eye problems later on in a person’s life. It may also occur alongside other conditions.
Potential eye-related complications
Other eye conditions people with aniridia might develop include:
- Cataracts – Cataracts are when a “cloud” or “spokes” form in the lens of your eye.
- Glaucoma – Glaucoma refers to a group of diseases that can damage the optic nerve, which sends information from the retina to the brain. It’s often caused by increased fluid pressure inside the eye (intraocular pressure).
- Nystagmus – Nystagmus is when your eyes move involuntarily, whether from side to side, up and down, or in a rolling motion.
- Keratopathy – Keratopathy refers to non-inflammatory diseases of the cornea. In aniridia-associated keratopathy, this might manifest as “clouding” of the cornea or scarring.
- Retinal hypoplasia – Hypoplasia refers to a lightened appearance to the retina. It can be related to symptoms of nystagmus.
- Microphthalmia – A person with microphthalmia is born with one or both eyes that are underdeveloped.
- Optic nerve anomalies – Defects of the optic nerve that may affect vision.
Other health concerns
Changes to the PAX6 gene could affect other body parts the gene is responsible for. This could lead to systemic conditions (ones that affect the whole body) like high blood pressure or diabetes. It could also lead to neurologic conditions (ones that affect the nervous system) like autism or epilepsy.
Other conditions that may appear alongside aniridia include:
- Wilms tumor – A cancerous kidney tumor (usually in just one kidney) found primarily in children. Like aniridia, Wilms tumor is congenital.
- WAGR syndrome – An acronym for its main features (Wilms tumor, aniridia, genitourinary abnormalities, range of developmental delays). WAGR syndrome tends to also involve issues relating to the kidney, pancreas, obesity or allergies.
How is aniridia diagnosed?
It’s possible to see the missing irises in a baby’s eyes after it’s born. Further tests conducted by an eye doctor will diagnose aniridia.
Eye exams
Aniridia can be diagnosed using common eye exam tests. Some of the tests that could diagnose aniridia include:
- Slit lamp exam – A slit lamp is a microscope your eye doctor uses to examine the cornea, iris, lens and pupil. For aniridia, this test can also be useful for assessing the risk of cataracts and glaucoma.
- Fundoscopy – A fundoscopy (or ophthalmoscopy) is when your eye doctor evaluates your eyes using light and magnifying lenses with or without dilation. Fundoscopy can be used to examine the retina and optic disc (the connection between the optic nerve and the retina).
- High-frequency ultrasound biomicroscopy – Ultrasound biomicroscopy creates an image of the eye using high-frequency sound waves. It can be especially useful for evaluating infants who cannot sit for a slit lamp or dilated fundoscopy examination.
These tests may be used to assess the severity of the condition and other affected structures in the eye. The doctor will also test visual acuity (sharpness of vision) at the initial visit and all follow-up exams. This helps your eye doctor see how your eyes might function in the future.
Genetic testing and counseling
If you’re concerned about the chances of your baby having aniridia, your eye doctor, primary care doctor or pediatrician may refer you to a genetic counselor for genetic screenings. They can estimate the baby’s chances of having aniridia by reviewing specific genetic and blood tests, along with samples of amniotic fluid, for the PAX6 mutation. These specialists play an important role in family planning and chronically managing aniridia.
If a baby does develop aniridia but the parents don’t have an obvious case of it, it’s recommended for the parents to undergo a comprehensive eye exam.
READ MORE: How DNA health testing can detect eye diseases
Is there treatment for aniridia?
Treatment for aniridia focuses on preserving vision and managing symptoms. Regular eye exams are important to keep on top of potential complications.
Vision support and aids
Glasses or contact lenses may improve vision for people with aniridia. There are even special colored contacts resembling an iris that can reduce light sensitivity. These can be prescribed by a contact lens specialist. Another option is tinted glasses or photochromic lenses that may help relieve light sensitivity.
Low vision aids can also be beneficial and prescribed by a low vision specialist. These include other types of lenses and assistive technology, like handheld telescopes and electronic devices that can magnify screens.
READ MORE: How to help someone who is visually impaired
Medical and surgical treatments
If your child develops a condition alongside aniridia, medical or surgical treatment may be needed. Their eye care provider may recommend iris implant surgery to possibly relieve light sensitivity, improve their ability to see and increase self-confidence.
Eye drops or artificial tears can assist with issues relating to the cornea (keratopathy). Medicated eye drops can be used to treat glaucoma. However, if eye drops do not improve either condition, then surgery may be required. Cataract surgery may be necessary if cataracts are severe.
Ongoing monitoring is essential to treat these conditions and their symptoms.
Other specialists who could be involved in the care of your child are:
- Pediatric oncologist – A specialist who focuses on diagnosing and treating different types of childhood cancers. With aniridia, patients need to be screened yearly until age 8.
- Urologist – A specialist who focuses on treating urinary diseases or disorders.
- Endocrinologist – A specialist who focuses on diagnosing and treating inflammatory or hormone conditions like diabetes or thyroid disease.
- Nephrologist – A specialist who focuses on diagnosing and treating kidney diseases.
- Pediatric ophthalmologist – A specialist who focuses on diagnosing and treating childhood eye conditions like strabismus (lazy eye). They may work with optometrists, glaucoma specialists, cornea specialists, retina specialists, neuro-ophthalmologists and cataract specialists when required.
Outlook for people with aniridia
Aniridia and its complications (like cataracts and glaucoma) can cause significantly reduced vision. Low vision aids are important for those with severe visual impairment and assisting with school work. It is also recommended to seek social services support.
Some organizations that provide resources for people with aniridia are:
When should I see my child's eye doctor?
You should consult an eye doctor if your child has any changes in their vision. Seek emergency care if:
- There is a sudden loss of vision.
- There are new floaters or flashing lights in their vision.
- Your child is experiencing eye pain.
Regular eye exams will be needed to monitor for conditions like glaucoma, cataracts and other eye problems.