Treatment for glaucoma
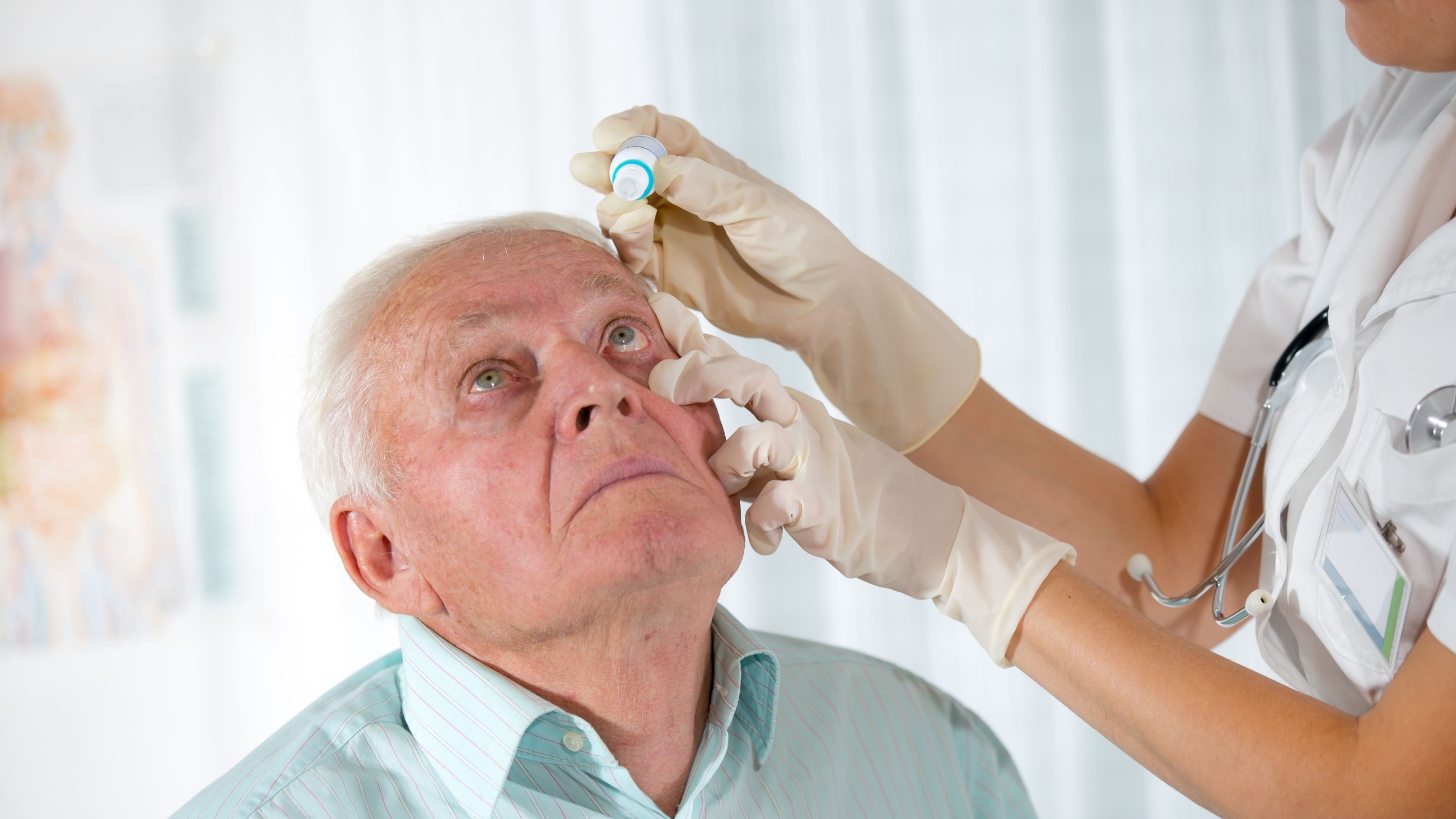
Glaucoma treatment works by lowering internal eye pressure. It usually begins with prescription eye drops that must be taken daily. Other possible treatments include oral medication, laser treatment and surgery. Glaucoma treatment plans are based on the type and stage of glaucoma a patient has.
Glaucoma is a group of eye diseases that can damage the optic nerve at the back of the eye. Optic nerve damage usually occurs because the pressure within the eye (called intraocular pressure or IOP) gets too high. If IOP stays high, the sensitive optic nerve fibers become damaged, which can result in vision loss or blindness.
When IOP starts rising, it's usually related to how easily aqueous fluid can travel through and then out of the eye. If the aqueous can't move freely, pressure starts to build up. So, all types of glaucoma treatment, in one way or another, focus on improving aqueous flow to lower IOP.
READ MORE about what causes glaucoma
Eye drops
The first step in glaucoma treatment is typically prescription eye drops. Your doctor may also call them ophthalmic solutions or topical solutions. They usually work by balancing the amount of aqueous fluid within the eye. Some of them help the eye’s drainage system work more effectively, and some reduce how much aqueous the eye produces.
There are many types of eye drops for glaucoma treatment. Each type falls into a category based on its active ingredients. The main active ingredient categories include:
- Prostaglandins
- Alpha agonists
- Beta blockers
- Rho kinase inhibitors
- Cholinergics (miotics)
- Carbonic anhydrase inhibitors
Some solutions also contain combinations of these drugs.
Topical solutions for glaucoma treatment can be very effective, but side effects are fairly common. Some patients may also have an allergic reaction to them.
Most side effects and allergy responses tend to be mild. However, some can be harder to tolerate. If you notice either of these, talk to your eye doctor right away about changing your drops, but don't stop using them.
Stopping your medication or not using it exactly as directed can lead to more vision damage. Your eye doctor can find a better alternative for you based on the types of effects you experience.
Allergic reactions are usually related to the type of preservative in the eye drop. Most topical solutions contain a preservative called benzalkonium chloride (BAK). People who are allergic to BAK may do better with drops that have a non-BAK preservative or are preservative-free.
Prostaglandins
Eye drops with this active ingredient have a teal lid or bottle cap color and work by increasing the level of fluid being filtered out of the eye.
This is done in two ways: First, they relax the ciliary muscle, which increases flow space for eye fluid. They also break down collagen and proteins to make more room between cells. This allows the fluid to circulate more easily.
Prostaglandin analogs are often an eye doctor’s first choice for treating open-angle glaucoma. They are effective for most patients, and they only need to be used once a day. Prostaglandins also have relatively fewer eye and vision side effects, though they are possible:
More common:
- Eye redness, itchiness and stinging
- Eyelash growth
- Darkening of skin around the eyes
- Changes in iris pigmentation
Rare:
- Ptosis (eyelid drooping)
- Blurred or double vision
- Photophobia (light sensitivity)
- Iridocyclitis (when the iris and ciliary body of the eye become inflamed)
- Hyphema (when blood collects in the front part of the eye)
You may also experience one or more systemic adverse reactions. Systemic means the effect presents itself in other areas of the body. These are generally caused by an allergic reaction to the drug and may include:
- Shortness of breath
- Skin rash or allergic dermatitis
- Nausea
- Chest pain
- High blood pressure
- Joint, bone or muscle pain
Types of prostaglandin analogs include:
- Tafluprost – (Zioptan™) Preservative-free; no generic available
- Travoprost – (Travatan® Z) Non-BAK; available in generic form
- Latanoprost – (Xalatan®, Xelpros™) Available in generic forms
- Bimatoprost – (Lumigan®) Available in generic form; (Durysta™ implant) No generic available
- Latanoprostene bunod – (Vyzulta™) No generic available
- Omidenepag isopropyl – (Omlonti®) No generic available
Latanoprost drops are usually the first glaucoma treatment doctors recommend. They can lower IOP by 30% or more for most patients within just a few months. Plus, they’re easier for patients to use properly and consistently since they’re a once-daily medication.
Durysta™
Durysta is a newer type of prostaglandin that was approved by the FDA for treatment of glaucoma in 2020. Unlike other prostaglandins, Durysta isn't a topical solution. It's a tiny implant that’s injected into the front of the eye with a special needle. The implant releases a steady dose of medication as it dissolves over the course of 90 days.
The Durysta implant can lower IOP for one to two years in most patients, but it can only be used once per eye. It may be a good option for those who don't do well with eye drops but also don't need glaucoma surgery yet.
Durysta’s side effects are similar to those of topical prostaglandin drops. However, there is an increased risk for macular edema and corneal tissue complications.
Alpha agonists
Antiglaucoma alpha agonists can reduce IOP by improving the eye’s ability to drain fluid and by reducing the amount of fluid it produces. Eye drops in this category have a purple lid and are often prescribed as treatment for open-angle glaucoma, but they can also be used to:
- Treat ischemic optic neuropathy (optic nerve damage caused by poor blood flow)
- Prevent ocular hypertension after laser eye surgery
- Treat or prevent complications associated with corneal transplants
- Reduce eye redness
When using alpha agonists, you may experience one or more eye-related side effects, such as:
- Eye burning or stinging upon application
- Allergic conjunctivitis
- Eye discharge or tearing
- Blepharitis (eyelid inflammation)
- Dry eye
- Corneal inflammation (keratitis) or damage (keratopathy)
Systemic effects may include:
- Dry mouth and/or nose
- Drowsiness
- Headache
- Fatigue
- Rash
- Indigestion
- Distortion in taste and smell
Types of alpha agonists include:
- Brimonidine tartrate – (Alphagan® P) Non-BAK; available in generic form
- Apraclonidine HCI – (Iopidine®) Available in generic form
Beta blockers
Beta blockers are frequently prescribed as treatment for glaucoma and ocular hypertension. Their bottles have a yellow cap (for 0.5%) or a blue cap (for 0.25%). Beta blockers work by decreasing the eye’s aqueous production, but they can also slow down the heart and circulatory system. Your doctor may avoid them if you have asthma, heart disease, kidney disease or diabetes.
The ciliary body contains protein molecules called beta receptors. When certain hormones stimulate these receptors, it triggers the ciliary body to make more aqueous. This medication works as a glaucoma treatment by blocking those receptors.
These drops are prescribed to be used once or twice per day. They usually have relatively mild eye and vision-related side effects, including:
- Reduced night vision
- Inflammation
- Blepharoconjunctivitis
- Blurry vision
However, beta blockers can also cause some serious systemic and eye-related issues. These include:
- Insomnia
- Dizziness and vertigo
- Headache
- Shortness of breath
- Low blood pressure
- Depression
- Reduced heart rate
- Heart block or failure
- Fatigue
- Impotence
- Abnormal blood vessel formation in the retina
- Cataracts
It may be possible to reduce systemic effects by taking certain steps during application. Close your eyes immediately after you apply the drops — but don't blink. Then lightly press your fingers over your tear ducts for one to two minutes. This can help keep the drug from entering your bloodstream via the tear ducts.
If you’re taking beta blockers for treatment of glaucoma and notice any of the symptoms listed above — especially dizziness, shortness of breath, tiredness or cold hands and feet — see a doctor immediately.
Types of beta blockers include:
- Levobunolol HCI – (Betagan®) Available in generic form
- Timolol maleate – (Timoptic-XE®, Timoptic® in Ocudose® PF, Istalol®) Available in preservative-free (PF) and generic forms
- Betaxolol HCI – (Betoptic® S) Available in generic form
- Timolol hemihydrate – (Betimol®) No generic available
Rho kinase inhibitors
Rho kinase inhibitors have been available since 2018. Eye drops with this active ingredient have a white cap and manage glaucoma in three ways:
- Increasing aqueous humor outflow
- Limiting eye fluid production
- Lowering pressure in episcleral veins
Episcleral veins are the tiny veins in the sclera (the white part of the eyes). Elevated pressure in these veins has a direct effect on intraocular pressure.
The drops have had great success in managing eye pressure and only have a few eye and vision-related side effects:
- Stinging or burning
- Conjunctival hyperemia (redness and dilation of vessels in the conjunctiva)
- Conjunctival hemorrhage
- Corneal deposits
Rho kinase inhibitors cause zero systemic reactions.
Types of rho kinase inhibitors:
- Netarsudil – (Rhopressa®) No generic available
Because rho kinase inhibitors are a new glaucoma treatment, there is no generic version yet. Some insurance providers may not cover them, which can make them an expensive option.
Miotic or cholinergic agents
Miotics, which usually have a green lid, are one of the oldest types of eye drops used as treatment for glaucoma. They reduce eye pressure by increasing aqueous drainage through the trabecular meshwork.
This is achieved when the miotics stimulate the parasympathetic nervous system and cause the pupils to contract. Contraction of the pupils allows for greater outflow of aqueous humor.
In addition to treating glaucoma, miotics may also be used to prevent ocular hypertension after eye surgery. They can be used alone or with other glaucoma treatments.
However, they are rarely used anymore due to the prevalence of reported side effects, including:
- Dim vision
- Brow achiness
- Cataracts
- Eyelid muscle twitching
- Uveitis (inflammation of the uvea)
- Cyst formation in the iris
- Corneal clouding
- Nearsightedness (more common in younger patients without presbyopia)
- Retinal detachment (rare)
Some systemic effects associated with miotics include:
- Slow or irregular heartbeat
- Fainting
- Increased urination
- Nausea and vomiting
- Diarrhea
- Sweating
Types of miotics include:
- Pilocarpine HCI – (Isopto® Carpine) Topical solution; available in generic form
- Carbachol – (Miostat®) Eye injection performed by a doctor
Carbonic anhydrase inhibitors
Carbonic anhydrase inhibitors (CAIs) are prescribed to treat both open- and closed-angle glaucoma. They stabilize eye pressure by decreasing the amount of fluid the eye produces. CAIs can be prescribed as pills or in topical form (which will have an orange lid).
The drops work by suppressing carbonic anhydrase, which is an enzyme found in ciliary body tissue. This enzyme is responsible for fluid secretion in many parts of the body, including aqueous humor. Suppressing carbonic anhydrase limits fluid secretion within the eye, reducing IOP.
CAIs for the treatment of glaucoma usually need to be taken twice a day. They are often paired with other antiglaucoma medications, such as beta blockers. Some systemic effects that have been linked to oral CAIs include:
- Memory issues
- Kidney stones
- Depression
- Upset stomach
- Numbness and strength loss of the hands and feet
- Frequent urination
- Having a metallic taste in your mouth
- Low potassium levels
CAIs can cause an electrolyte imbalance in some people. Before starting CAIs for glaucoma treatment, tell your doctor if you have kidney disease or blood electrolyte issues.
If you use topical CAIs, you may experience one or more of the following:
- Eye burning or stinging
- Eyelid crusting
- Blurred vision
- Light sensitivity
- Double vision
- Corneal inflammation or damage
Types of carbonic anhydrase inhibitors include:
- Acetazolamide – Oral (open-angle) or intravenous (acute angle-closure); only available in generic form
- Methazolamide – Oral; only available in generic form
- Dorzolamide HCI – (Trusopt®) Topical solution; available in generic form
- Brinzolamide – (Azopt™) Topical solution; available in generic form
Combination eye drops
Some people require a combination of medications for glaucoma treatment. Combination eye drops contain two or more medications in a single solution, which makes it easier to stay on track with your dosing schedule.
The lid color of combination drops may be blue, white or light green, depending on the compound of medication. Combination drops are more convenient and potentially less expensive than using multiple medications. Plus, using fewer medications may reduce your exposure to irritating preservatives.
However, they also come with some possible drawbacks. Because they contain two or more active ingredients, you may experience the side effects of each one.
Before starting a combination prescription, talk to your eye doctor about the potential side effects of each active ingredient.
Types of combination drops include:
- Carbonic anhydrase inhibitor + beta blocker – (Cosopt®, Cosopt® PF) Available in preservative-free (PF) and generic forms; blue bottle cap
- Alpha-adrenergic agonist + beta blocker – (Combigan™) Available in generic form; blue bottle cap
- Alpha-adrenergic agonist + carbonic anhydrase inhibitor – (Simbrinza®) No generic available; light green bottle cap
- Rho kinase inhibitor + prostaglandin analog – (Rocklatan®) No generic available; white bottle cap
Many people have a tough time remembering to use their drops, or they skip doses because of side effects. This is understandable, but glaucoma medications simply won't work without diligent use.
Glaucoma is usually a chronic condition. Most patients require lifelong treatment, including using eye drops every day. Failing to do so increases your risk of vision loss.
Never discontinue or skip doses of your medication without first consulting your doctor. They will work with you to find alternative glaucoma treatments.
Oral medicines for glaucoma treatment
Sometimes, topical solutions alone aren't enough to reach a patient's target IOP. In these cases, your doctor may prescribe oral medication to replace or supplement them.
Oral medications prescribed for the treatment of glaucoma are usually a type of carbonic anhydrase inhibitor. Two common types are methazolamide and acetazolamide.
Methazolamide may be taken two or three times daily to manage glaucoma. Common eye-related and systemic effects include:
- Vision changes
- Numbness or tingling in your extremities
- Drowsiness
- Taste changes
- Ringing in the ears
- Loss of appetite
- Confusion
- Nausea and vomiting
Acetazolamide is a tablet that may be taken one to four times daily, depending on your doctor’s recommendations. Systemic effects that you may experience while taking this medicine include:
- Dizziness
- Fatigue
- Headache
- Ataxia (impaired coordination)
- Confusion
- Hypoglycemia (low blood sugar)
- Anaphylaxis (severe allergic reaction)
- Seizures
When discussing glaucoma treatment options with your eye doctor, be sure to mention important health details. Details may include medications, vitamins or supplements you are taking. These details can help your doctor recommend the best and safest glaucoma treatment for you.
Surgeries
There are several different types of glaucoma surgery. Some involve the use of lasers or minimally invasive incisions. Others are more complicated with more invasive techniques. The different types of surgery treat different types and stages of glaucoma, but they all have the end goal of lowering eye pressure.
If eye drops and oral medications are not working to lower your IOP enough, you may need glaucoma surgery. You may also need surgery if you aren't able to use your drops exactly the way your doctor says you should.
Surgical glaucoma treatment can significantly lower IOP. But often, it's necessary in addition to your other medications. It’s important to understand that many people need to keep using eye drops after surgery.
The best type of glaucoma surgery for each patient depends on many factors, including:
- Age
- Type of glaucoma
- Other underlying conditions
- Level of eye pressure
- Previous eye surgery
Laser trabeculoplasty
Laser trabeculoplasty is a procedure used to treat open-angle glaucoma and, in rare cases, angle-closure glaucoma. During the procedure, the surgeon uses a laser that targets the trabecular meshwork.

Click on illustration to enlarge.
The goal of the surgery is to adjust the porous space within the trabecular meshwork to allow the aqueous fluid to flow more freely. This encourages more outflow of aqueous humor, which lowers IOP.
Laser trabeculoplasty only takes about five to 10 minutes, and its effects can last two or three years. Typically, patients can return to their normal routine the day after surgery.
Laser peripheral iridotomy
This procedure is commonly performed on patients with angle-closure glaucoma. A laser peripheral iridotomy involves using a laser to puncture a tiny hole in the iris. Doing this creates an “escape hatch” for aqueous humor to drain from the eye, which helps balance IOP.

Click on illustration to enlarge.
Peripheral iridotomy can also be performed without using a laser. Rather than using a laser, the surgeon uses a needle with a tip that bends at a 45-degree angle. The needle is inserted into the eye, behind the iris, and used to create a puncture in the iris. Microsurgical scissors are then used to remove a small piece of iris tissue. This procedure takes around five minutes for each eye.
After a peripheral iridotomy, you’ll need to have your eye pressure checked frequently (every 30-60 minutes). You may have blurred vision and some mild discomfort or feel like something is in your eye. These are temporary and will resolve within a few hours to a few days.
Laser cyclophotocoagulation
In laser cyclophotocoagulation, a surgeon uses a laser to target a specific area of the ciliary body called the ciliary processes. Generally speaking, this procedure can be used to treat all forms of glaucoma.
The surgery takes about 10 minutes to complete, and the goal is to shrink the ciliary processes using the laser. This makes the ciliary processes produce less aqueous humor and helps stabilize IOP. A potential downside of this procedure is that the ciliary processes regenerate, so the procedure will need to be repeated several times.
Because this procedure involves damaging the ciliary processes, it’s often reserved for when other glaucoma surgeries are unsuccessful or for other specific cases.
Scatter panretinal photocoagulation
While this procedure is often used to treat retinal disease, it has also been successful in treating neovascular glaucoma (NVG). In NVG, new blood vessels grow into the drainage angle of the eye and clog it. Panretinal photocoagulation uses a laser to keep these abnormal blood vessels from growing.
The surgery takes around 15-30 minutes to complete. You may notice eye discomfort or achiness after the procedure, as well as dim vision. This is normal and should resolve shortly after surgery.
Trabeculectomy
Trabeculectomy is considered the gold standard of surgical treatment for glaucoma. It is typically reserved for advanced cases, but it can treat nearly any type of glaucoma. The goal of a trabeculectomy is to create a new pathway for eye fluid to drain, which helps balance IOP.

Click on illustration to enlarge.
During a trabeculectomy, the surgeon creates a flap in the white area of the eye (sclera). This is usually done in an area of the sclera that’s covered by the upper eyelid. Then the doctor makes a pathway from inside the flap into the front of the eye. The flap is then put back in place, and the entire surgical site is covered by the eye’s conjunctiva. The length of the procedure is one to two hours.
The results of the surgery include a small, blister-like bubble on the surface of the flap. This is called a “bleb,” and the fluid in it comes from the new pathway. The bleb is hidden under the eyelid, and the fluid it collects is absorbed back into the body.
Canaloplasty
The eyes have a natural drainage system called the canal of Schlemm. This canal collects aqueous humor from the trabecular meshwork, then transports it to the veins of the eyeball to be absorbed.
A canaloplasty is a procedure in which a tiny catheter is run through the canal of Schlemm. Once in place, the catheter expands to help widen the canal. With a wider canal, aqueous humor should be able to drain more effectively, and IOP levels should balance out.
It takes anywhere from 45 minutes to two hours to perform a canaloplasty. The surgery has shown success in treating open-angle glaucoma and congenital glaucoma.
Drainage Implant surgery
Drainage implants (also called aqueous shunts or tube shunts) are a common surgical treatment for glaucoma. They are used for many forms of the disease, including congenital and childhood glaucoma.

Click on illustration to enlarge.
There are different kinds of drainage implants that may vary slightly. However, they all consist of the same features: a soft, flexible tube that attaches to a small plate. The plate is designed to match the curvature of your eyeball, so it rests against it comfortably.
Surgery to install the implants is also generally the same. Your eye surgeon will create a tiny opening under the conjunctiva. They will then insert the plate in the opening, where it rests on top of the sclera. The tube that’s attached to the drainage plate is embedded into the anterior chamber (front part) of your eye.
Aqueous humor will flow out through the tube and collect in the plate. As fluid collects, a bleb (the blister-like bubble mentioned earlier) may form. Rest assured; the fluid will be naturally reabsorbed. Typically, the entire surgery takes an hour or less to complete.
Trabecular bypass stent
This procedure has proven to be a successful treatment for glaucoma. However, it is usually only performed in conjunction with cataract surgery. It takes about 15 minutes to complete (not including the time it takes to perform the cataract surgery).

Click on illustration to enlarge.
Implanting a trabecular bypass stent is similar to a canaloplasty in that it focuses on Schlemm’s canal. But rather than inserting a catheter, the surgeon places tiny implants made of titanium through the canal.
The ophthalmologist uses a needle-like injector to enter the eye and then navigates it to Schlemm’s canal. The injector is preloaded with two stents, which allows for two implantations within a single entry to the eye. Once in place, the stents allow for better aqueous flow through the canal, which helps lower IOP.
Deep sclerectomy
Some surgeons view a deep sclerectomy as a safer alternative to trabeculectomy for the treatment of glaucoma. This is because a trabeculectomy penetrates the anterior chamber, which creates potential for complications.
In a deep sclerectomy, the surgeon creates a scleral flap that is deep enough to reach Schlemm's canal. Then they remove the top layer of the canal, or its "roof," as well as some of the trabecular meshwork.
They also remove some scleral tissue from the underside of the flap to create a space between the sclera and the canal. A tiny implant is placed into this new space to keep it open, and then the flap is put back into place. The new space then acts as a sort of internal bleb to collect and filter aqueous fluid.
A deep sclerectomy takes between 30 and 45 minutes to complete. The surgery has been used to successfully treat primary glaucoma and secondary glaucoma due to uveitis.
Minimally invasive glaucoma surgeries (MIGS)
Minimally-invasive glaucoma surgeries (MIGS) are micro-surgery techniques developed to reduce or eliminate the need for glaucoma medications. MIGS require only tiny incisions and cause less trauma to the eye than conventional glaucoma surgery. However, many MIGS are only done at the time of cataract surgery.
The goal with MIGS and conventional glaucoma surgery is the same: to increase outflow of aqueous humor from the eye. Doing so helps lower IOP and decrease the risk of optic nerve damage and vision loss.
Glaucoma procedures that fall under MIGS:
- Trabecular stent bypass
- Canaloplasty
- Deep sclerectomy
- Laser cyclophotocoagulation
- Types of trabeculotomy procedures, like gonioscopy-assisted transluminal trabeculotomy (GATT) and the Kahook Dual Blade (KDB)
Cataract surgery
While cataract surgery alone has been shown to reduce IOP by over 16%, it is usually not relied on to treat glaucoma. However, it’s common for the two conditions (cataracts and glaucoma) to coincide with one another.
For this reason, it’s possible that your eye doctor may recommend a combined surgery that treats both conditions in a single procedure. Some perks of having combined surgery include fewer risks, lower cost and increased convenience.
It’s important to note that combined surgery isn’t for everyone. Before your eye doctor approves a combined surgery, they will assess certain details about your condition. These details include:
- The type and advancement of your glaucoma
- Whether medication alone is successfully managing your IOP
- The severity of your cataracts (whether they’re interfering with the requirements of daily life)
If they determine you’re a good candidate, you may be scheduled for a combined procedure. Combined surgery often includes some method of cataract removal and a MIGS procedure.
Marijuana and CBD
Marijuana and other cannabis-derived substances like CBD are often discussed as options for treatment of glaucoma.
Though it’s true that marijuana can lower eye pressure, the effect it has is temporary. In people with glaucoma, eye pressure must be managed 24 hours a day. This makes marijuana an impractical treatment for glaucoma. The amount and frequency of marijuana use required would be extremely expensive and cause a number of unwanted side effects.
Further, marijuana could actually harm the optic nerve more than help it. The optic nerve can be damaged by high IOP, but it can also be damaged by insufficient blood flow. Marijuana lowers blood pressure in addition to IOP. For many people, the danger marijuana presents to the optic nerve could outweigh its benefits as a treatment for glaucoma.
Prevention
There is currently no cure for glaucoma.
However, glaucoma can usually be managed successfully with early detection and regular treatment. By treating glaucoma early and diligently, vision loss can be minimized or prevented in most cases.
The best treatment for glaucoma is prevention — catching the disease before it causes noticeable vision loss. It’s important to note that open-angle glaucoma (the most common type) doesn’t have symptoms. This makes prevention all the more important for ensuring eye health.
Some simple ways to help prevent glaucoma include:
- Learning your family history of eye disease – Most glaucoma is hereditary, so finding out whether the disease runs in your family is a great place to start in glaucoma prevention.
- Having regular comprehensive eye exams – Whether you’re considered high risk or not, a comprehensive eye exam can detect signs of many diseases, including glaucoma.
- Controlling your blood pressure – High blood pressure has a direct effect on your eye pressure. Keeping your blood pressure in check is important for your vision and overall health.
- Practicing a healthy lifestyle – Being physically active, incorporating eye-healthy foods into your diet and maintaining a healthy weight can help with glaucoma prevention, as well as other eye diseases.
- Avoiding smoking – Smoking has zero health benefits, including the health of your eyes. If you want to prevent the negative effects smoking has on your eyes and vision, ditch the habit.
- Protecting your eyes from injury and UV exposure – Injury to the eye can cause glaucoma to develop later on — a condition called traumatic glaucoma. Similarly, damage from sun exposure can lead to several eye conditions, like cataracts, macular degeneration and glaucoma. Protecting your eyes with safety glasses or sunglasses can help lower your risk.
- Avoiding unnecessary pressure on the eyes – Certain sleeping positions where the pillow or another object puts pressure on one or both eyes aren’t ideal. Exercises or activities that require you to be head-down, like yoga, can also raise eye pressure. If possible, try to avoid putting or raising pressure in the eyes unnecessarily.
- Talking to your doctor about current medications and health conditions – Whether it’s an oral or topical steroid or medicine for hypertension, talk to your doctor about your current prescriptions and how they may affect your eye health. And don’t forget to mention the health conditions discussed above to your doctor.
When to see an eye doctor
Any changes in your vision, like blurred, faded or darkened peripheral vision, should be checked out by an eye doctor. If you are using prescription medication or eye drops for glaucoma treatment, it is vital that you attend all of your scheduled check-ups.
Patients who have had any type of glaucoma surgery must also attend all their scheduled follow-up appointments with the eye surgeon.
Treatments for glaucoma can prevent vision loss, but they require diligence and your eye doctor’s guidance.
Amber McManes also contributed to this article.