Understanding AMD
Age-related macular degeneration (AMD) is a progressive eye disease affecting central visual acuity. It is among the leading causes of vision loss in older adults. Understanding the risks to your vision and getting treatment when needed can help you manage the condition and preserve your sight.
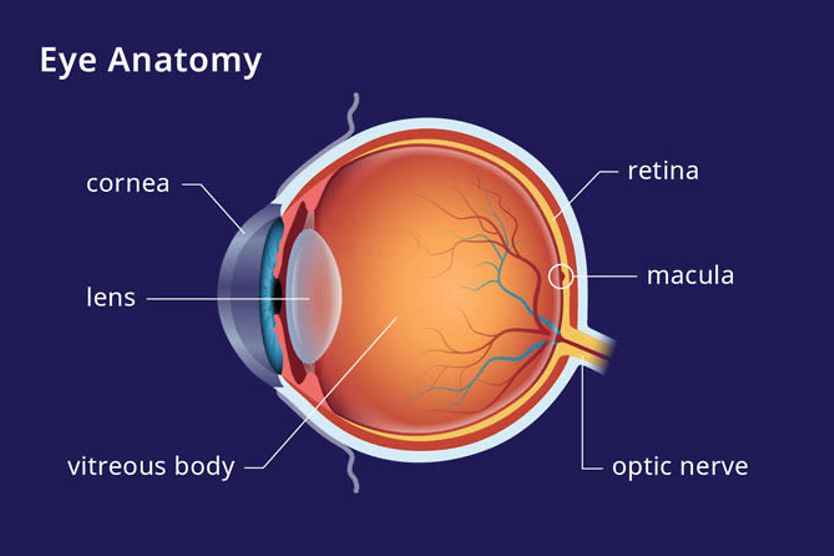
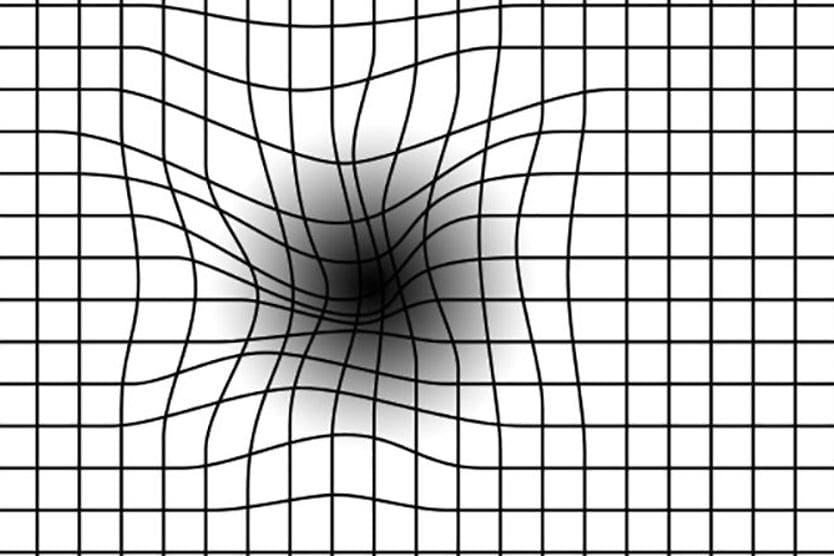
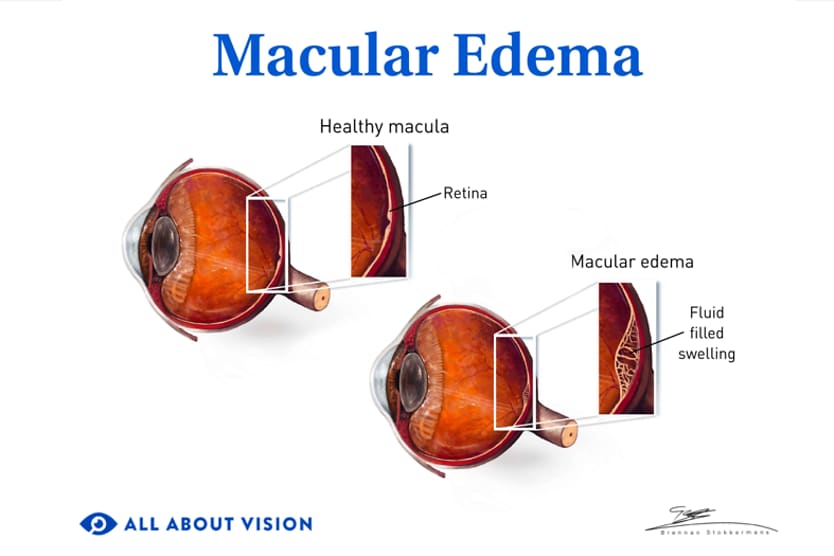
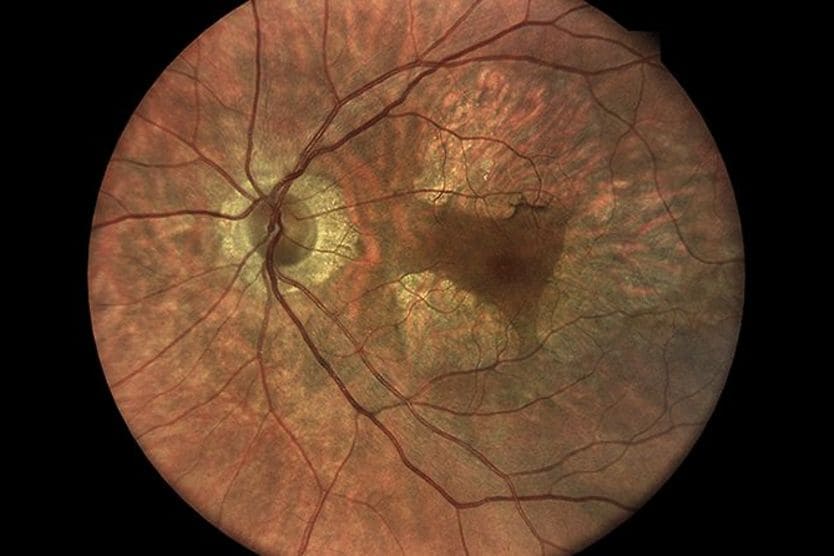
AMD results from age-related damage to the macula — the center part of the retina that helps maintain your sharpest vision. When this occurs, your central vision may become blurred, wavy or distorted. (Your central vision allows you to see what is directly in front of you). Other symptoms, such as blind spots or areas of missing vision, may also develop.
Macular degeneration does not affect peripheral (side) vision. It can occur in one or both eyes.
AMD eye disease can affect your life in many ways. It can impact your ability to read, drive and perform other everyday tasks. If the disease progresses to the advanced stages, there may be a substantial loss of central vision.
Types of AMD
There are two types of age-related macular degeneration, known as dry AMD and wet AMD. Almost all cases begin as the dry variation of the disease.
Dry AMD is the most common form of the condition. It generally progresses slowly, causing a gradual loss of central vision. Dry AMD is associated with the thinning and breakdown of the macula. The presence of yellow deposits in the macula, called drusen, also occurs with this form. Drusen are made up of proteins and lipids.
There are three stages of dry macular degeneration: early, intermediate and advanced. Dry AMD may progress to an advanced stage known as geographic atrophy (GA). GA can lead to severe vision loss.
Dry AMD is also called atrophic, non-neovascular and nonexudative macular degeneration.
Wet AMD is less common than the dry form but typically causes vision loss to occur at a faster rate. It is an advanced stage of AMD eye disease. Wet macular degeneration occurs when abnormal blood vessels develop in or beneath the retina and leak blood or other fluid. This process can scar and damage the macula. A protein known as vascular endothelial growth factor (VEGF) causes this vessel growth and leakage.
Wet AMD is also called exudative or neovascular macular degeneration.
There is no cure for wet or dry AMD. However, certain treatments can slow AMD progression. Treating macular degeneration can help preserve your sight and possibly prevent additional vision loss.

Click on image to enlarge.
Common treatments for macular degeneration
The options for treating macular degeneration depend on the type of AMD present.
Eating a balanced diet that contains nutrient-rich foods may be helpful to those with AMD. These foods include fish, yellow vegetables and fruits, and spinach, kale or other leafy greens.
SEE RELATED: How to boost your diet to protect aging eyes
Medications for macular degeneration
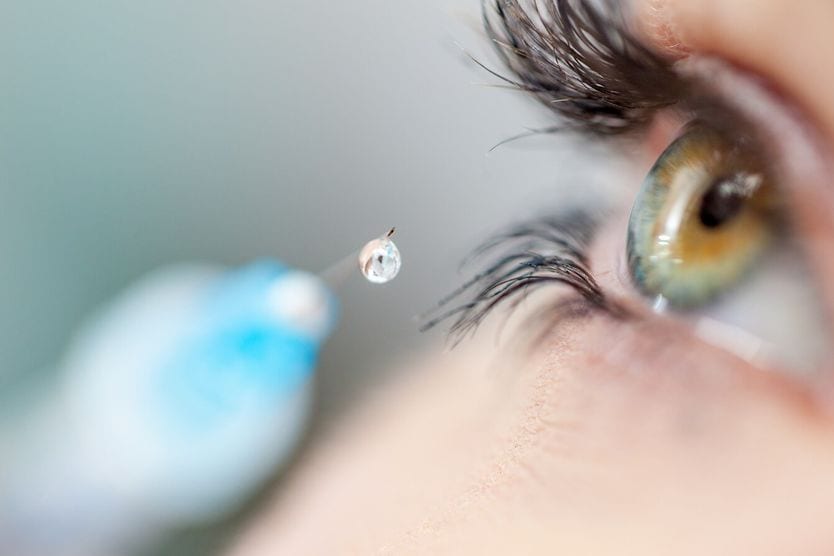
Medications for macular degeneration are given in the form of intravitreal eye injections. They are injected into the jelly-like vitreous fluid located in the center of the eye. Eye injections may be used to potentially inhibit the progression of advanced forms of the disease. They may be given periodically to help manage advanced dry AMD and wet AMD.
Dry AMD treatment options
Early dry macular degeneration treatment
There is no current medical-based treatment for dry macular degeneration in the early stage. Nutritional intervention is the only potential therapeutic option at this time. Eating a diet rich in antioxidants may slow the progression of dry AMD. Following the Mediterranean diet may also reduce the risk of early-stage dry AMD.
Intermediate dry macular degeneration treatment
The National Eye Institute sponsored two studies known as the Age-Related Eye Disease Studies (AREDS and AREDS2). These studies involved patients with cataracts and age-related macular degeneration. They found that taking certain formulations of nutritional supplements may reduce the risk of AMD progression.
The first AREDS study revealed that taking a dietary supplement containing the following reduced the risk of serious AMD progression by 25%:
- Vitamin C
- Vitamin E
- Zinc
- Copper
- Beta-carotene
The second study was called AREDS2. It was conducted to see if changes to the original AREDS formula might provide further benefits against vision loss from advanced AMD. The results showed that adding lutein and zeaxanthin to the original formula lowered the risk of progression to advanced stages of AMD.
*Beta-carotene was also removed from the formula. High doses of this supplement have been linked to an increased risk of lung cancer in smokers.
The AREDS2 formula may be recommended for people with the early to intermediate stage of dry AMD. It includes specific antioxidant vitamins and minerals combined into a capsule form and taken orally. These vitamins and minerals include:
- Vitamin C (500 mg)
- Vitamin E (400 IU)
- Zinc (80 mg)
- Copper (2 mg)
- Lutein (10 mg)
- Zeaxanthin (2 mg)
Taking these supplements may prevent further progression of the condition. It may also reduce the risk of developing wet AMD. An eye doctor can determine if the AREDS2 formula may be helpful for a particular stage of the disease.
Certain vitamins, minerals or other supplements may interfere with some medications. Speak with your eye doctor or health care professional before taking nutritional supplements.
SEE RELATED: How to choose eye and vision supplements
Advanced dry macular degeneration treatment (for geographic atrophy)
Until recently, there were no treatments available for advanced dry AMD (geographic atrophy). Two medications have recently received FDA approval for advanced dry macular degeneration treatment:
- Pegcetacoplan – Pegcetacoplan received FDA approval for the treatment of geographic atrophy in February 2023. It is the first drug to ever receive approval for treating GA. This macular degeneration medication is injected into the eye every 25-60 days. It may slow the progression of geographic atrophy. These injections are given monthly or once every other month.
- Avacincaptad pegol – Avacincaptad pegol received FDA approval for the treatment of GA in August 2023. It is also injected into the eye and works to possibly slow the progression of geographic atrophy. These injections are given monthly or once every other month.
Treatments for wet macular degeneration
Treating wet macular degeneration involves the following factors:
- Inhibiting the growth of new blood vessels beneath or within the retina
- Reducing or preventing the leakage of these vessels
Some wet AMD treatments may slow the progression with the goal to potentially preserve and stabilize your vision as much as possible.
Anti-VEGF injections (anti-angiogenic drugs)
The most common treatment for wet macular degeneration is anti-VEGF medications. These medications are also known as anti-angiogenic drugs. Vascular endothelial growth factor (VEGF) is a protein involved in the development of abnormal blood vessels. This happens through a process called angiogenesis.
Injected into the eye, anti-VEGF medications help prevent the development of new abnormal blood vessels in the retina. They also help prevent the leakage from these blood vessels that gives “wet” macular degeneration its name.
Imaging techniques (such as optical coherence tomography) and exams to evaluate visual acuity are performed before any of these treatments and during follow up visits. The results of these exams are used to determine how frequently future doses should be given.
Several types of macular degeneration eye injections are currently used in the treatment of wet macular degeneration. Some of the most common include:
- Ranibizumab – Ranibizumab became FDA-approved for the treatment of age-related macular degeneration in 2006. It is administered once a month to the affected eye.
- Bevacizumab– Bevacizumab is approved by the FDA to treat various types of cancer but has been used “off-label” in the treatment of wet AMD since 2005. This macular degeneration injection is typically given every four to six weeks. However, the dosing schedule can vary based on individual needs.
- Aflibercept – Aflibercept, also called “VEGF trap-eye,” was originally approved by the FDA for wet AMD treatment in 2011. In August 2023, a higher-dose regimen also received FDA approval. The first three doses are administered to the affected eye once per month. Additional injections are given every two to four months.
- Faricimab-svoa – Faricimab-svoa received FDA approval for the treatment of wet AMD in January 2022. It is given as an eye injection once per month for the initial four doses. Additional treatment can be given every one to four months in the affected eye.
- Brolucizumab-dbll – Brolucizumab-dbll received FDA approval for the treatment of wet AMD in October 2019. This medication for macular degeneration is administered once per month for the first three months. Follow-up doses are given every two to three months.
Biosimilar anti-VEGF medications may also be used to treat wet AMD. A biosimilar drug is highly similar biologically to that product's original or brand-name version. A generic drug, on the other hand, is an exact chemical copy of the original product.
Like the original version, a biosimilar must undergo testing and FDA approval, which can be costly. However, some of the information from the original testing process can be used to develop a biosimilar. This means they can often be made more quickly and possibly at a lower cost. For these reasons, they may offer cost savings to some patients.
Two biosimilar anti-VEGF drugs have received FDA approval for the treatment of wet AMD. These include:
- Ranibizumab-nuna – Ranibizumab nuna received FDA approval in September 2021. It is given as a monthly eye injection.
- Ranibizumab-eqrn – Ranibizumab-eqrn was approved by the FDA in August 2022. It is also given as an eye injection once per month.

For the "wet" form of macular degeneration, medication is carefully injected into the eye.
AMD injections: Benefits and risks
The use of anti-VEGF injections for macular degeneration treatment can be effective for many cases of wet AMD. The benefits of this treatment option can vary according to the type of macular degeneration injection used. AMD injections may:
- Improve visual acuity
- Stabilize central vision
- Slow the development of new blood vessels in or beneath the retina
- Hinder or slow the advancement of retinal damage from abnormal blood vessels
- Slow the progression of vision loss from AMD
Like many medications used to treat medical conditions, anti-VEGF eye injections may carry risks or cause some side effects. These could include:
- Increased intraocular (eye) pressure
- Eye pain
- Eye infection
- Bleeding within the eye
- Eye floaters
- Retinal tear
- Retinal detachment
- Cataract
- Changes in vision
Side effects may vary according to the type of drug administered or other factors. Speak with your eye doctor to understand the benefits and risks of treating wet AMD with anti-VEGF injections. It is important to notify your eye doctor should you experience any side effects.
Eye injections for macular degeneration: Procedure and aftercare
Eye shots for macular degeneration are administered by an ophthalmologist. The procedure generally involves the following steps:
- Before the procedure begins, the intraocular pressure and visual acuity of the affected eye are evaluated.
- The patient will be partially reclined with their head and neck supported by a headrest.
- The surface of the eye is cleansed with an antiseptic solution.
- The eye is then numbed to enhance treatment comfort.
- The eyelids are also cleansed with an antiseptic solution.
- A special device (called a lid speculum) is used to keep the eye open during the procedure.
- A drop of povidone-iodine 5.0% is applied to disinfect the designated injection site.
- The medication is injected through the white of the eye (commonly the lower, outer area) while the patient looks in the opposite direction. The injection only takes a few seconds to complete.
- Following the injection, the patient’s overall visual acuity is checked once again.
- The lid speculum is removed, and a saline solution is used to rinse the eye.
Aftercare instructions will be provided by the ophthalmology team. It is common for your eye to be sore for a couple of days following the injection. Your vision may also be blurry, and you may notice eye floaters. The use of antibiotic drops may be recommended for the first one to two days following treatment.
For the first several days following eye injections, you may be advised to avoid:
- Rubbing your eyes
- Swimming
- Wearing makeup
- Gardening
- Working in a dusty environment
Other treatments for macular degeneration
In addition to eye injections, other macular degeneration treatment options may be available to some patients.
Implantable telescopes
This device is designed for people with severe vision loss due to end-stage (advanced) wet or dry AMD. Around the size of a pea, it is surgically placed in the eye to replace the natural lens. It magnifies images onto the healthy parts of the retina, reducing the size of blind spots in central vision caused by AMD. The device was approved by the FDA in 2010.
A second generation of the device was developed in 2020. Smaller in size, it requires a smaller incision and less surgical time to place. These factors may offer additional benefits.
Photodynamic therapy (PDT)
PDT is a treatment for macular degeneration that combines a drug called verteporfin and a low-intensity laser. It targets leaky, abnormal blood vessels affecting the macula in wet AMD. PDT was approved for use by the FDA in 2000.
During this process, verteporfin (a light-sensitive drug) is administered into the arm intravenously. The drug makes its way through the body to the vessels in the eye. The eye is then numbed with a special eye drop. A contact lens is placed on the eye, and the laser is projected through this lens into the back of the eye to activate the drug. This process causes clots to form in the abnormal vessels, possibly closing them off.
PDT is not used as frequently as it was in the past due to the increased use of anti-VEGF medication. However, it may also be used in addition to anti-VEGF injections.
Laser photocoagulation
Laser photocoagulation is another type of laser treatment for macular degeneration. It was the first treatment used to address wet AMD. The procedure uses laser light to destroy or seal off new abnormal blood vessels to prevent leakage. Since it is only ideal for specific types of retinal damage, only a certain percentage of people are candidates for this procedure.
Before the procedure begins, the eye is numbed for treatment comfort. A special contact lens is then placed on the eye. The doctor shines a laser light on the retina, in the back of the eye, to target and close off abnormal vessels.
A drawback of laser photocoagulation is that it can scar the macula. This may cause blind spots in a person’s visual field. Laser photocoagulation is not as widely used for treating AMD due, in part, to the risks of the treatment and the increased use of anti-VEGF drugs.
LEARN MORE about laser photocoagulation
Alternative and emerging treatments
Over the last 10 to 15 years, medical advancements have brought about sight-saving outcomes with the use of anti-VEGFs for wet AMD. However, these options require routine applications to potentially maintain vision stabilization or improvements. Medicine-based treatment options for early dry AMD are also yet to be confirmed.
The development of longer-lasting anti-VEGF drugs for treating macular degeneration continues to be underway.
Ongoing research and clinical studies are being conducted to identify new treatments for both dry and wet AMD. Learn more about current clinical studies in macular degeneration by visiting ClinicalTrials.gov.
Innovative approaches to AMD treatment
Many of the efforts to develop new treatments for macular degeneration involve replacing eye injections with other options. These include:
Port delivery system
The port delivery system is a newer option designed for the treatment of wet AMD. It involves an anti-VEGF injection delivered through an implant placed within the eye. The implant’s reservoir releases medication over a certain period of time. This treatment aims to reduce the need for periodic eye injections.
Some implant delivery systems for the sustained release of medications have shown promise. However, further research and development are needed before this treatment can be widely offered to AMD patients.
Stem cell research
Researchers are currently looking at stem cell therapy as a macular degeneration treatment option. The idea behind stem cell therapy involves replacing retinal cells destroyed by dry AMD with healthy stem cells. Research in the use of stem cell therapy for both wet and dry AMD is underway.
Gene therapy
Gene therapy for macular degeneration treatment is also currently under investigation. This treatment would encourage the eye to generate its anti-VEGF medications on its own or create new retinal cells. Gene therapy is delivered via a single eye injection. The idea is that one injection could provide the patient with long-term, lifetime results. Gene therapy is being researched to target forms of both dry and wet AMD.
Eye drops
The use of eye drops is another potential AMD treatment option. Further clinical trials are needed to assess the safety and efficacy of this approach.
Oral medications
Various types of oral medications are being studied for the treatment of wet and dry AMD. These include:
- An oral anti-VEGF to replace eye injection treatments for wet macular degeneration
- An oral antibiotic, doxycycline, for the treatment of geographic atrophy
- Metformin, a diabetes drug, to reduce inflammation and other factors associated with macular degeneration
Combination drug treatments
Combination drug therapies are being studied for wet macular degeneration treatment. The goal is to see if giving two drugs at once can improve vision and provide longer-lasting results.
Living with age-related macular degeneration
Being diagnosed with AMD can be challenging for an individual and their loved ones. Living with AMD often centers on preserving their quality of life. It also involves finding ways to make the most of their remaining vision. This can mean learning to use their peripheral vision along with other senses. Adjusting to these changes typically takes some practice.
Low vision specialists can help identify ways to optimize a person’s remaining visual function. They may also suggest the use of low vision devices to carry out a variety of daily tasks. These may include magnifiers, glasses or electronic options.
The importance of early detection
Macular degeneration can affect a person’s visual function to varying degrees. Severe forms of age-related macular degeneration could even lead to legal blindness. The potential effects of this condition make early detection and treatment very important.
There is no cure for age-related macular degeneration. However, treatments for AMD can help improve the quality of life for those affected by the disease. Seeing an eye doctor as recommended is essential to help monitor the condition. Eye exams are also important in preventing AMD and detecting it as early as possible.
READ NEXT: Warning signs of age-related eye problems