What is diabetic retinopathy?
Diabetic retinopathy (DR) is a serious eye disease that can affect people with diabetes mellitus. It occurs when high levels of glucose (sugar) in the blood cause changes to the network of blood vessels located in the retina. These changes can damage the retina, leading to vision loss and blindness.
Diabetes is a condition characterized by the chronic elevation of glucose levels in the blood. In the body, blood sugar levels are controlled by insulin. When there is too little insulin produced or the body is resistant to insulin, diabetes may result. Diabetes can impact many organs and systems of the body, including the eyes.
The retina is the light-sensing membrane located in the back of the eye. It converts light and visual images into special signals. These signals are then sent to the brain to process what you see.
Changes to the retina, such as those that occur from diabetes, can impact its ability to function properly. These changes can result in issues that lead to visual impairment.
Diabetic retinopathy is the most common eye condition among individuals with diabetes. It is also one of the top causes of blindness in the U.S. adult population. Diabetes-related retinopathy affects 93 million people worldwide.
SEE RELATED: Other types of retinopathy
Causes of diabetic retinopathy

Click image to enlarge.
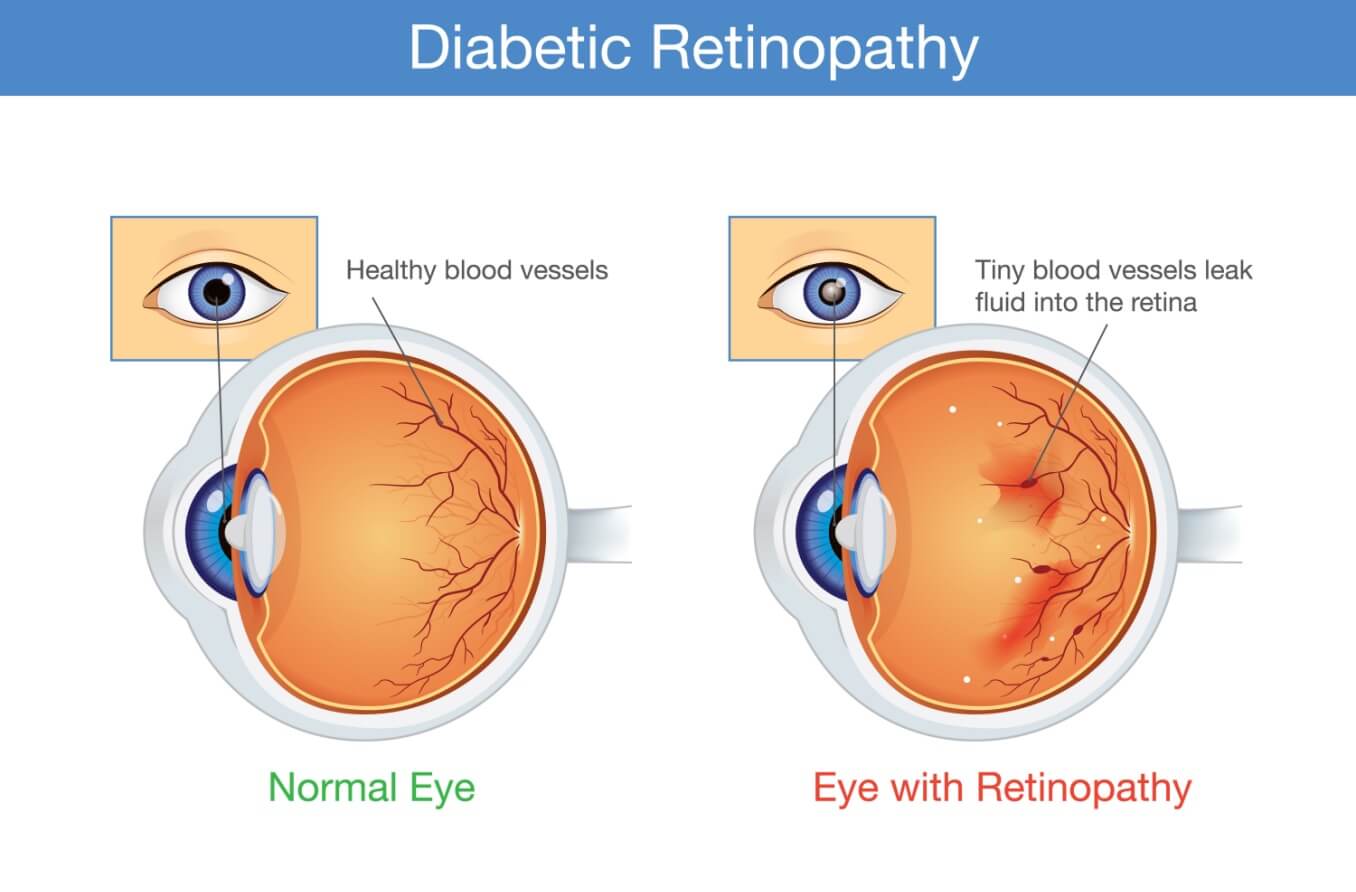
Several factors may lead to the development of diabetic retinopathy. However, prolonged hyperglycemia (high blood sugar levels) is thought to be the leading reason behind its onset.
High blood sugar can cause the blood vessels in the retina to become enlarged and leak fluid. In other cases, the growth of new, abnormal vessels in the retina may occur. At times, the vessels in the retina may become blocked. This prevents blood flow through this part of the eye, depriving it of oxygen and nutrients. Each of these factors can damage the blood vessels in the retina, causing diabetic retinopathy.
Who is at risk?
Diabetic retinopathy can affect people with Type 1 or Type 2 diabetes. Women who develop pregnancy-related diabetes (gestational diabetes) may also be at risk. DR may occur in those with both diagnosed and undiagnosed diabetic conditions.
The longer you have diabetes, the higher your risk of developing DR.
The biggest risk factors include:
Poorly controlled blood sugar levels
Long-term diabetes
High cholesterol
High blood pressure
Smoking
Pregnancy
Age (50 and over)
Family history of diabetes
Obesity
Ethnicity may also increase a person’s risk. People within the following populations may be more likely to develop DR:
Hispanic/Latino
African American
American Indian
Alaska Native
Signs and symptoms
The early stage of DR may not cause any symptoms. When diabetic retinopathy symptoms do occur, however, they may include:
Eye pain or pressure
Eye redness
Poor night vision
Changes in color vision
Partial or total vision loss
People with this condition typically experience symptoms in both eyes.
READ MORE: Blurry vision and diabetes
What is the first sign of diabetic retinopathy?
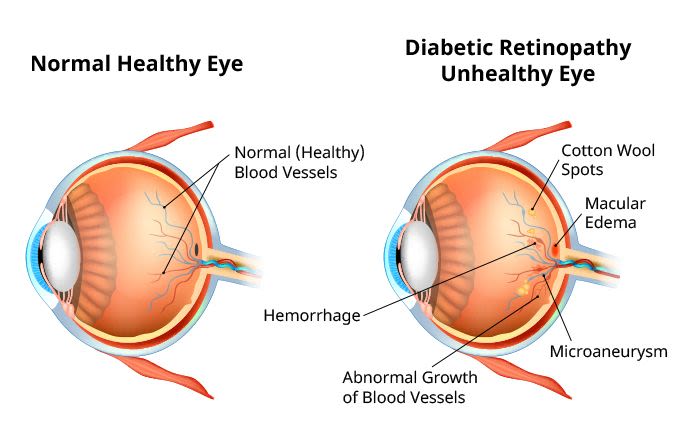
One of the first signs of diabetic retinopathy is the presence of microaneurysms in the blood vessels of the retina. These are bulges within the vessels that leak fluid or blood. They often look like small red dots sprinkled across the retina when viewed through diagnostic equipment.

Microaneurysms are a characteristic feature of diabetic retinopathy. Other signs include:
Blood in the retina (retinal hemorrhage)
Thickening of the macula (macular edema)
Fatty deposits (hard exudates) in or under the retina
Cotton wool spots (soft exudates) in the retina
As the condition progresses, further changes to the retina may develop. These include:
Abnormal vessels and vascular changes within the retina
New vessels growing within the retina or iris
Blood in the vitreous (vitreous hemorrhage)
Scarring of the retina
The blockage or loss of macular blood vessels, depriving the macula of nutrients and oxygen (macular ischemia)
READ MORE: Types of diabetic eye problems
Main types and stages of diabetic retinopathy
Diabetes-related retinopathy is classified into two primary types. These include non-proliferative diabetic retinopathy and proliferative diabetic retinopathy. The main difference between the two is whether new, unhealthy blood vessels have developed in the retina.
The condition is further categorized into four stages of diabetic retinopathy progression.
Type: Non-proliferative diabetic retinopathy (NPDR)
NPDR is the early stage of the general condition. It occurs in three stages of progression — mild, moderate and severe. The key factors in this stage are leaky retinal vessels and blockages in small blood vessels called capillaries. This leads to swelling, inflammation and tissue damage in the retina.

Click image to enlarge.
Stage 1: Mild non-proliferative diabetic retinopathy
Mild NPDR is the earliest stage of the condition. A few microaneurysms are generally present at this stage of the disease.
Stage 2: Moderate non-proliferative diabetic retinopathy
In this stage, the number of microaneurysms and areas of bleeding in the retina increases. Doctors refer to these areas of bleeding as dot-and-blot hemorrhaging. Other retinal changes may also arise. These include the presence of fatty deposits and cotton wool spots.
Increased swelling of the blood vessels may occur. The vessels may also become blocked.
Stage 3: Severe non-proliferative diabetic retinopathy
The condition is in the severe NPDR stage when the following factors are present:
Microaneurysms and scattered hemorrhaging occur within all four quadrants (quarters) of the retina.
Venous beading (where the vessels take on a sausage-link appearance) is present in at least two quadrants of the retina.
Intraretinal microvascular abnormalities (IRMA) develop in at least one quadrant of the retina.
Doctors refer to this as the “4-2-1 rule.”
The noticeable effects of non-proliferative diabetic retinopathy may include:
Blurred vision
Blind spots
Gradual loss of vision
NPDR can eventually trigger the growth of new blood vessels in the retina (neovascularization). It can also lead to diabetic macular edema (DME).
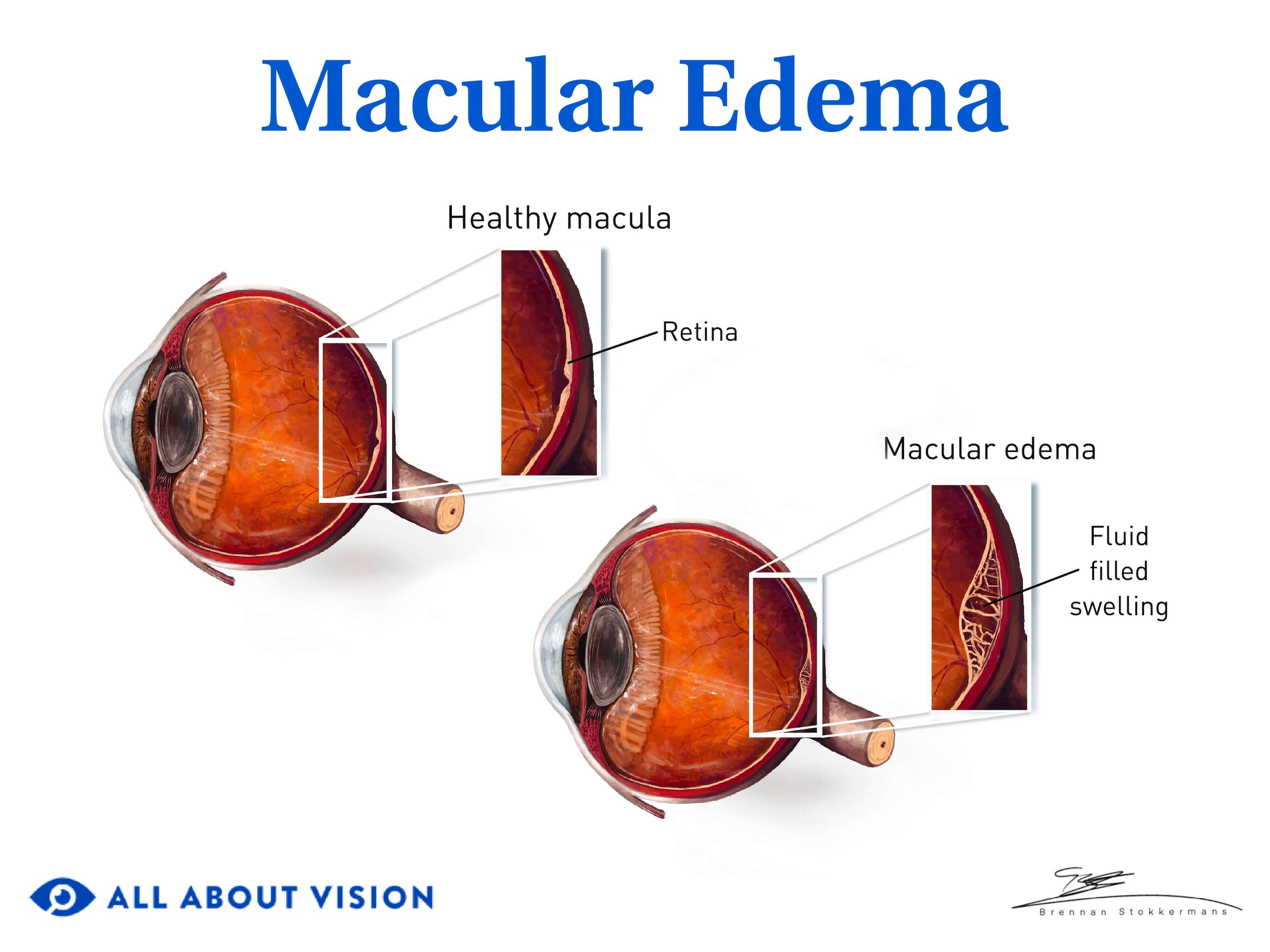
Diabetic macular edema
DME is swelling and thickening of the macula and is the leading cause of vision loss in people with DR. It can develop during any DR stage and may require treatment, even if it appears in mild NPDR.
Neovascularization occurs during the advanced stage of DR. This stage is called proliferative diabetic retinopathy.
SEE RELATED: What are Roth spots?
Type: Proliferative diabetic retinopathy (PDR)
PDR is characterized by the development of new but abnormal blood vessels in the retina.
Stage 4: Proliferative diabetic retinopathy
When normal blood vessels in the retina become blocked or damaged, the body tries to find another way to help blood reach these tissues. These changes trigger the release of a protein called vascular endothelial growth factor (VEGF), which aids in the creation of new (but abnormal) vessels. This is called neovascularization and indicates the fourth stage of the disease.
The development of abnormal vessels can cause scar tissue to form. The scar tissue can pull or tug on the retina, leading to a condition called retinal detachment.
The vessels may also leak, allowing blood to enter the vitreous (the gel-like fluid in the back of the eye). Known as vitreous hemorrhage, this can prevent light from reaching certain points on the retina. Vitreous hemorrhage can impact your vision, from causing a mild increase in floaters to completely blocking out your vision. It can clear up by itself over time, but its cause will always require some type of treatment.
Proliferative diabetic retinopathy may lead to further eye conditions, including:
Neovascular glaucoma
Diabetic macular edema
Other effects of proliferative diabetic retinopathy may include:
Eye flashes or floaters
Distorted vision
Sudden loss of vision
Significant vision impairment or blindness
Diabetic macular ischemia
Diabetic macular ischemia (DMI) is a common complication of diabetic retinopathy. It most often presents in advanced cases but may develop at any DR stage.
DMI occurs when blood vessels in the macula become narrowed or blocked, deteriorate or are destroyed. This impedes blood flow into the macula, depriving it of necessary oxygen and nutrients. DMI can cause mild to severe vision loss, depending on its location and severity.
Diagnosis, management and treatment
The earlier diabetic retinopathy is detected and treated, the better the chances of keeping it from progressing. Recognizing early stage diabetes eye symptoms can lead to timely treatment and management of the condition. Your doctor may be able to detect signs of DR even before you have symptoms.
How is the condition diagnosed?
Diagnosing diabetic retinopathy typically begins with a comprehensive eye exam. Your eye doctor may use the following tests to diagnose the disease:
Visual acuity exam – This exam uses eye charts to test your ability to see objects clearly at different distances.
Amsler grid test – This test uses a square grid made of straight lines, rather than a chart with letters. It allows the doctor to assess your central vision for distortions or blind spots.
Dilated retinal exam – Your doctor may place drops in your eyes to temporarily dilate (widen) your pupils. This allows them to see into the back part of your eye, where the retina sits.
Tonometry – Tonometry measures eye pressure (intraocular pressure) and is used to screen for glaucoma. It may be part of the exam since people with diabetes have a higher risk of developing glaucoma.
Ophthalmoscopy – With this technique, your doctor uses a special device called an ophthalmoscope. This device has magnifying lenses used to examine the retina.
Fundus photography – Your doctor may take pictures of the fundus (back of the eye) to check the retina and other structures.
Fluorescein angiography – Fluorescein (a yellow dye) is administered into a vein. The dye moves through your bloodstream and into the vessels in your eyes. A special camera is then used to take pictures of the retina as the dye moves through the vessels. With this exam, your doctor can determine if new blood vessels have developed. It also allows them to see any areas of blood vessel blockage or leakage.
Optical coherence tomography (OCT) – Using light wave technology, OCT produces cross-sectional images of the eye’s structures. These images allow your doctor to view the layers of the retina to check for changes or damage. It also measures the thickness of the retinal layers.
In some instances, artificial intelligence (AI) and teleophthalmology are used to screen for DR. Teleophthalmology is a form of telemedicine. It allows doctors to deliver eye care through digital methods. Medical providers can use these tools to detect signs of the condition remotely, which could lead to an earlier diagnosis.
SEE RELATED: Retinal imaging and scans
Regular eye exams are essential to detect diabetic retinopathy vision problems early and prevent further vision loss. Your doctor may use the results of your eye exam to determine if or which diabetic retinopathy treatments are needed.
Diabetic retinopathy treatment is commonly provided by a retinal specialist, which is an ophthalmologist specializing in the retina.
What treatment options are available?
Patients with mild diabetic retinopathy generally do not need treatment. At this stage, controlling blood sugar and blood pressure, and blood cholesterol and lipid levels is key to managing the condition.
Your eye doctor may recommend one or more of the following treatments for advanced cases of DR:
Laser
Laser surgery closes off blood vessel leaks and reduces retinal swelling (macular edema). These procedures may also help shrink abnormal blood vessels and reduce the risk of retinal detachment.
There are two main types of laser treatment for diabetic retinopathy. These include:
Focal or grid laser photocoagulation – This treats certain cases of macular edema. It involves focusing laser energy directly on the affected area or in a grid-like pattern on the retina. Focal laser treatments target specific microaneurysms. Grid laser treatments address larger areas of blood vessel leakage.
Scatter (panretinal) laser photocoagulation – Advanced cases of DR, or proliferative diabetic retinopathy, may be treated with panretinal photocoagulation (PRP). This is a more rigorous type of laser diabetic retinopathy surgery. Laser surgery with PRP is designed to reduce the amount of VEGF produced by the retina. In this way, it lowers the risk of new, abnormal vessel development (neovascularization).
Laser diabetic retinopathy treatments are generally performed in an ophthalmologist’s office or an outpatient treatment center.
SEE RELATED: Laser coagulation eye surgery
Vitrectomy
A vitrectomy involves surgically removing some or all of the vitreous fluid from the eye.
This may be recommended in advanced cases of diabetic retinopathy, such as when blood has leaked into the vitreous gel and is impairing vision. The vitreous cavity is then filled with a clear fluid to help maintain the shape of the eye.
A vitrectomy may also be conducted if scar tissue has formed in the retina.
Vitrectomies are typically performed at a hospital or an outpatient surgery center.
Anti-VEGF eye injections
Eye injections of medications known as anti-VEFGs can help stop the overproduction of VEGF in the eye. They help inhibit the growth of abnormal blood vessels and shrink those that have already developed.
Anti-VEGFs are injected into the vitreous of the eye. Along with preventing blood vessel growth, these eye injections help control macular swelling. They also reduce the risk of vessel leakage and retinal scarring.
Anti-VEGF medications used for DR are often the same ones utilized for macular degeneration injections. Some of the more common medications used for diabetic retinopathy include:
Eylea (aflibercept)
Lucentis (ranibizumab)
Avastin (bevacizumab)
Eye injections are provided in your ophthalmologist’s office. They may be recommended on a monthly basis, depending on the type of medication used and other factors. Your eye doctor will tell you how often you should have diabetic retinopathy medication injections.
Corticosteroids
Corticosteroid medications may also be used as a diabetic retinopathy treatment. Steroids reduce fluid leakage and swelling in the retina to help control macular edema.
These diabetic retinopathy medication treatments may be given in the form of an eye injection on a monthly basis. As an alternative, an intraocular implant may be surgically placed in the eye. The implant is filled with a steroid such as dexamethasone or fluocinolone acetonide. Steroid implants may offer a longer-acting option over monthly eye injections.
Corticosteroid implants are generally used in cases of DR that are resistant to anti-VEGF injection therapy. They provide a controlled release of medication over a certain period of time.
Intravitreal steroid eye injections are performed in your ophthalmologist’s office. Implants may also be placed in your eye doctor’s office. In more complex cases, implant surgery may take place in a hospital or surgical center.
Can diabetic retinopathy be reversed?
The very early stage of diabetic retinopathy may be reversed with tight blood sugar control. Eye injections and surgery may reverse certain effects of the condition.
In some cases, treatment can help improve vision. But in many instances, vision loss is permanent, and the goal of treatment is to prevent additional vision loss
Yearly diabetic eye exams allow your doctor to detect the condition as early as possible. Early diagnosis and treatment can reduce the risk of disease progression.
How is the disease managed?
While diabetic retinopathy cannot be cured, you can take steps to manage the disease and help keep it from advancing.
Diabetes is a chronic, lifelong condition that can cause significant health issues if it is not controlled. Managing this disease through diet, exercise and medical care (including vision care) can help preserve your health and protect your eyesight. Controlling diabetes can slow the development and progression of diabetic eye diseases, such as retinopathy.
Other factors key to managing diabetic retinopathy include:
Controlling blood sugar levels – This is central to managing both diabetes and diabetic eye conditions. Be sure to eat a healthy diet and take diabetes medication or insulin as prescribed by your doctor to control glucose levels.
Controlling high blood pressure and blood lipids – Ensuring your blood pressure and blood lipids (such as cholesterol and triglycerides) remain under control can lower your chance of developing retinopathy.
Managing diabetes-related kidney problems – Treating kidney or other health issues caused by diabetes is essential to maintaining the condition.
Getting yearly diabetic eye exams – Having regular eye exams allows your doctor to detect signs of retinal diseases or other problems caused by diabetes. The earlier DR is detected, the better the chances of preserving your vision.
See your eye doctor for more on managing diabetic retinopathy and its effects.
Health risks and complications
Diabetic retinopathy can threaten your vision and cause visual impairment. It can also increase the risk of other eye conditions. However, the risks and complications of the disease affect health factors that go beyond your eyesight.
Diabetes-related retinopathy can also increase the risk of other health problems. Some of these include:
Cardiovascular (heart) disease
Stroke
Diabetic peripheral neuropathy (nerve dysfunction in the extremities)
Diabetic nephropathy (kidney dysfunction)
Diabetic foot syndrome
You could experience a range of other complications due to diabetes. Eye problems related to the disease can involve one or more of the following conditions:
Tractional retinal detachment
Neovascular glaucoma
Macular edema
Vision loss or blindness

There may be other medical problems associated with DR. Consult your doctor to learn more about your personal risks or health concerns.
Prevention
Some of the steps aimed at managing DR are the same used to help prevent it. It might not always be possible to prevent the onset of diabetic retinopathy. However, you may be able to reduce the risk of disease progression and vision loss. Factors that may lower these risks include:
Getting yearly eye exams – Having your eyes examined at least once every 12 months allows your doctor to detect and treat problems as early as possible. In fact, many people first learn they have diabetes through a routine eye exam.
Manage diabetes – Controlling diabetes is key to lowering the risks associated with DR. It is important to monitor and control your blood sugar and take diabetes medication or insulin as prescribed by your doctor. A healthy diet and proper nutrition are also important for controlling blood sugar.
Manage high blood pressure and blood lipids – Controlling your blood pressure and blood lipid levels can help lower the risk of diabetic retinopathy.
Get eye exams during pregnancy – If you are pregnant and have diabetes, get your eyes examined during each trimester. Inform your eye doctor if you develop gestational diabetes, as this is also a risk factor. Your eye doctor may recommend more frequent eye exams within the first year after giving birth.
Follow a healthy diet – Eating a healthy, balanced diet (such as the Mediterranean diet) may lower your risk for diabetes-related eye conditions. Certain vitamins and minerals may also help support your eye health. Talk to your doctor before taking any new dietary supplements.
Get regular exercise – Exercising regularly may lower the risk of eye diseases such as diabetic retinopathy. Physical activity also helps lower blood glucose levels in the body.
Reduce ultraviolet (UV) exposure – Prolonged exposure to UV rays can damage the eye’s tissues, including the retina. Reducing the amount of time spent in the sun may help lower your chance of developing DR.
Avoid tobacco use – Some studies have found that smoking may increase the risk of diabetic retinopathy. Quitting or avoiding smoking is important for your eye health and general wellness.
Speak with your eye doctor for further details on lowering your risk of diabetic eye disease.
When to call an eye doctor
Diabetic retinopathy is a serious eye condition that needs prompt care from an eye care professional. If you have symptoms of diabetic retinopathy or notice vision changes, visit an eye doctor right away. Contact your doctor to schedule an exam or find an eye doctor near you if you do not yet have one. Prompt care can help prevent vision loss and maintain your eyesight.
READ NEXT: 8 ways to protect your eyesight