Corneal cross-linking (CXL) is a minimally invasive outpatient procedure designed to treat progressive keratoconus (and, sometimes, other conditions that cause a similar weakening of the cornea).
The corneal cross-linking procedure can strengthen and stabilize the cornea by creating new links between collagen fibers within the cornea. The two-step procedure applies specialty formulated riboflavin (vitamin B2) eye drops to the surface of the eye immediately followed by a controlled exposure of the eye to ultraviolet light.
The two basic types of corneal cross-linking are:
- Epithelium-off cross-linking. In this procedure, the thin outer layer (epithelium) of the cornea is removed to allow the liquid riboflavin to more easily penetrate the deeper corneal tissue.
- Epithelium-on cross-linking. In this procedure (also called transepithelial cross-linking), the protective corneal epithelium is left intact.
Candidates for corneal cross-linking
Corneal cross-linking can be effective if it can be performed before the cornea has become too irregular in shape or there is significant vision loss from keratoconus. If applied early, cross-linking typically may stabilize or even improve the shape of the cornea, often resulting in better visual acuity and an improved ability to wear contact lenses.
Other potential off-label applications in the United States of cross-linking include the treatment of corneal ulcers that are unresponsive to treatment with topical antibiotics. Studies also have found that corneal cross-linking can be effective for eradicating a variety of corneal infections.
Some refractive surgeons outside the U.S. already offer corneal cross-linking as a simultaneous add-on to LASIK or PRK to strengthen the cornea and potentially reduce the risk of complications like post-surgical ectasia. While this combined approach is not yet FDA-approved in the United States, it has been studied internationally in patients with higher prescriptions or thinner corneas, and some U.S. surgeons perform it off-label. Standardized protocols and long-term data are still evolving.
Some surgeons have also reported results in improving the stability of the cornea in people who have undergone radial keratotomy (RK), an incisional refractive procedure from the 1980s and 1990s. Cross-linking appears to work for those who are experiencing daily fluctuations as a result of their RK procedures.
What to expect during and after corneal cross-linking
During a preliminary evaluation, your eye doctor will measure the thickness of your cornea and make sure you are a good candidate for the procedure. You also will need to have a routine comprehensive eye exam to assess your vision and general eye health.
Detailed mapping of the shape of your cornea (called corneal topography) will also be performed.

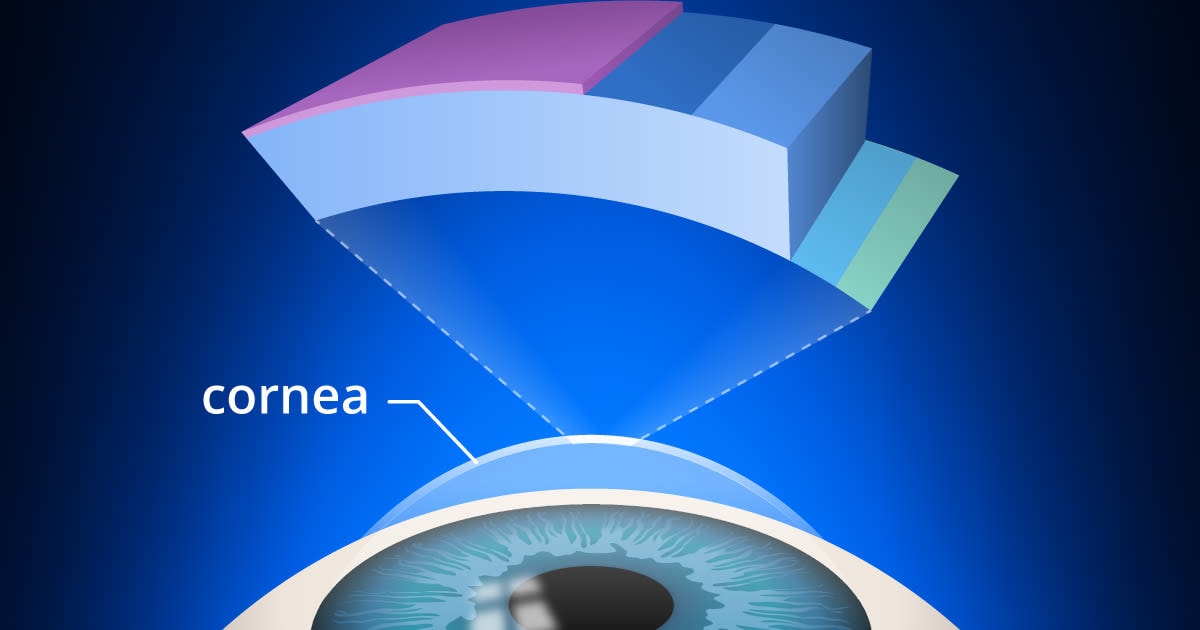
Layers of the cornea. Corneal cross-linking strengthens bonds in the stroma of the cornea, which is the layer from which tissue is removed during LASIK surgery.
The cross-linking procedure takes about an hour in many cases, depending on the technique used.
If you are undergoing an epithelium-off ("epi-off") procedure, your surgeon will place you in a reclining position and remove the epithelial layer from your cornea. Riboflavin eye drops will then be placed on your eye.
If you undergo an epithelium-on ("epi-on") procedure, you are placed in a reclining position, and riboflavin drops are applied. After enough time has passed (depending on the technique), the surgeon may evaluate your eye to ensure that the cornea has absorbed a sufficient amount of riboflavin.
Corneal thickness may be checked, and then the UV light will be applied for up to 30 minutes.
After epi-off and epi-on cross-linking, a bandage contact lens will often be placed on your eye to reduce discomfort and facilitate healing. Topical antibiotic and anti-inflammatory drops will be prescribed.
Epi-off cross-linking studies have found that many keratoconus patients can remain stable or achieve improvement in the corneal shape, which can be significant for people with progressive keratoconus that otherwise might lead to severe vision loss.
There is one device that is FDA-approved for epi-on cross-linking while another is currently under clinical investigation to determine the safety and efficacy of the procedure.
Cost of corneal cross-linking
Insurance coverage for FDA-approved corneal cross-linking is available through most major commercial plans, though coverage requirements and out-of-pocket costs vary. Your eye doctor's office or insurance provider can help you understand what to expect.